



UK Poultry Disease Quarterly Surveillance Report : January - March 2009
Highlights of the latest quarterly report from the Veterinary Laboratories Agency (VLA) include low pathogenicity avian influenza at two turkey premises, erysipelas and atypical necrotic enteritis in layers and severe gangrenous dermatitis in broilers.
 January – March 2009 Published June 2009 Contents OVERVIEW - FACTORS INFLUENCING DISEASE AND SUBMISSION RATES - NOTIFIABLE DISEASES - FARM VISIT INVESTIGATIONS - FOOD SAFETY INCIDENTS - ZOONOSES CROSS-SECTOR AVIAN DISEASES ENDEMIC DISEASE SURVEILLANCE |
Highlights
- Isolation of H6N1 low pathogenicity avian influenza (LPAI) viruses confirmed following investigation of two turkey premises - The incursion of H6 viruses into domestic poultry occurs sporadically in the EU. This incident demonstrates the potential for the incursion and rapid spread of virus within and between flocks.
- Erysipelothrix rhusiopathiae septicaemia causing very high cumulative mortality was seen in free-range layers - There is currently no licensed vaccine for use in chickens in the UK.
- Atypical necrotic enteritis was diagnosed in commercial layers - This is an unusual manifestation of this condition.
- Severe gangrenous dermatitis causing sudden onset high mortality in broiler chickens, led to suspicion of notifiable disease - Classical lesions were present in all birds. Gangrenous dermatitis is now very uncommon in broiler chickens.
Notifiable Disease - Great Britain
Domestic poultry
No outbreaks of avian notifiable disease (AND) were confirmed during the quarter (January to March 2009) in Great Britain. Clinical material was however submitted to the National Reference Laboratory (NRL) for Avian Influenza (AI) and Newcastle Disease (ND), VLA Weybridge from four cases of suspected AND in domestic poultry. Two of these investigations were in turkey flocks (located in Norfolk and Suffolk), and two in chicken flocks (located in Kent and Gwent). In total, 200 samples were tested comprising sera (60), oropharyngeal swabs (60), cloacal swabs (60) and carcasses (20). The isolation of H6N1 low pathogenicity avian influenza (LPAI) viruses was confirmed following investigation of the two turkey premises – further details of these cases are provided below.
H6N1 LPAI infection in turkey breeders - Case report
Introduction
Disease associated with H6N1 LPAI infection was confirmed affecting two separate turkey breeder premises during February 2009. The diagnosis was made as a result of investigations that were initiated following the reported suspicion of AND affecting both premises. All of the birds at each site were housed in fully bird-proofed (netted) pole barns. The premises are both located in Eastern England, are 'shower in-shower out' sites with strict biosecurity protocols, and are owned by the same company. No other company premises were affected.
History & Clinical signs
At the time of initial AND investigations, Site A comprised two turkey breeder flocks (Shed 1 and Shed 2) aged 50-weeks and 49-weeks-old respectively, with approximately 4,800 hens placed in each. Two further sheds of 250 stags each were also present. Site B comprised four turkey breeder flocks of approximately 2,500 hens each, aged 36-weeks, and two houses of some 500 stags. The duration and severity of disease was different on each premises, with recorded production losses and a more marked clinical presentation evident on Site A, approximately seven days before a clinical signs were observed at Site B. The constellation of observed clinical signs at Site A, coupled with the appearance of similar disease signs on a second premises within a short time frame prompted the PVS to report suspected AND on 24 February 2009.
The pattern of clinical signs and production losses in affected sheds at both sites was, however, broadly similar. Initially, an egg drop was recorded, followed within 24 to 48 hours by the onset of mortality and coughing, and a rapid clinical progression and resolution over a seven- to 10-day period. Within affected sheds, the birds were also consistently described as being quieter and lethargic, and clinically affected birds were initially reported to be at one end only, with increasing morbidity, progressing as a wave of affected birds throughout the house over the subsequent two- to four-day period. In addition to the recorded egg drops, loss of shell pigment was observed, with an increased number of floor eggs. Coughing was consistently reported to resolve within 72 hours of onset. However, lethargy, mortality and egg drop persisted for longer, most notably at Site A. Egg peritonitis and ‘pneumonia’ were observed at post-mortem examination of affected birds. At Site A, clinical signs were first evident in Shed 2 on 18 February 2009 (18 per cent egg drop in 24 hours). Prior to this, bird losses were attributable to culls, and egg production had been consistent (mean daily production of 3,300 eggs). In Shed 1, the onset of clinical signs was reported to be on 21 February (11 per cent egg drop in 24 hours). Figure 10 and Figure 11 show the daily egg drop and mortality data for Shed 2 and Shed 1 respectively.
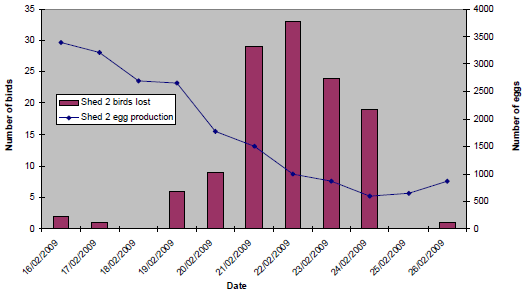
(16/02/09 - 26/02/09)
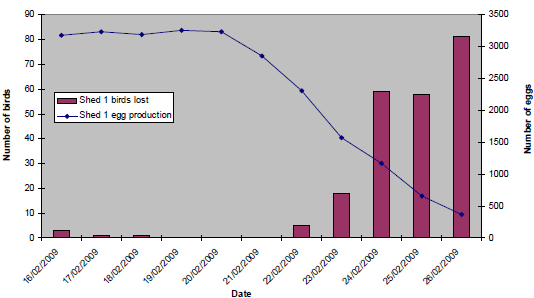
(16/02/09 - 26/02/09)
At Site B, clinical signs were evident in Shed 5 only from 21 February (onset of mortality), with a 16 per cent egg drop recorded by 24 February (previous mean daily production of 2,260 eggs). The overall severity and duration of production losses, clinical signs and mortality was much less at Site B (Figure 12).
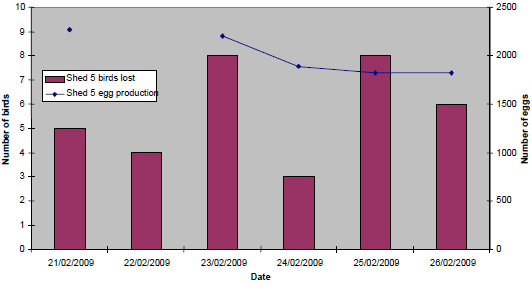
(21/02/09 - 26/02/09)
Laboratory Investigations
Samples comprising sera (40), oropharyngeal swabs (40), cloacal swabs (40) and carcasses (8) were submitted to the NRL for AI and ND, VLA Weybridge from birds in the affected sheds at both premises. Results from the influenza A virus Matrix gene RRT-PCR (positive), and the H5 and H7 RRT-PCRs (both negative) were available within 12 hours of the start of testing (on 25 February), consistent with the presence of non-notifiable (non-H5, non-H7) influenza A virus infection. Virus isolation (in embryonated fowls’ eggs) and further characterisation resulted in the identification of the H6N1 virus. Virus pathotype was confirmed as LPAI by intravenous pathogenicity index (IVPI) testing, resulting in the lifting of restrictions on both premises. No further statutory disease control measures were implemented.
In addition to the standard laboratory tests, results of initial genetic sequencing of the haemagglutinin (HA) gene of the H6 viruses isolated from both cases revealed 99 per cent HA gene similarity, being most closely related to other contemporary European H6 viruses for which data is available. Furthermore, whole genome analysis revealed, for all gene segments, closest similarity to viruses circulating in wild birds and poultry in the EU. The genotype of the UK isolates was not consistent with those H6 strains that have been reported recently in Asia.
Summary & Conclusions
The incursion of H6 viruses into domestic poultry occurs sporadically in the EU. Detection is most frequent in turkeys, presumably as a result of increased susceptibility to AI viruses and evident clinical presentation, and ducks, possibly due to frequent contact with wild waterfowl in which H6 viruses are not uncommon. In Great Britain from 2002-2008, eight H6 isolates were found through routine AIWBS activities. Whilst the source of the H6N1 LPAI virus was not definitively established, it is relevant that H6 isolates from both premises shared 99 per cent HA gene similarity, indicating either secondary spread from an index premises (putatively Site A), or a shared, but unidentified, common source exposure of both sites. A linear distance of some 30 miles separated the two affected premises, and no other flocks of the same type within the company were affected. Furthermore, HA gene and whole genome sequencing analyses confirmed the virus to be most closely related to other known, contemporary, European H6 viruses of poultry and wild bird origin. Based on the exclusion of other relevant epidemiological factors, it is therefore tempting to speculate that the virus may have been of wild bird origin.
This episode also demonstrates a number of epidemiological features relevant to AI. Firstly, the potential for the incursion and rapid spread (both within and between flocks) of avian influenza viruses, and the requirement for high levels of on-farm hygiene and biosecurity measures to be maintained. Secondly, the overall pattern of disease was similar at both affected premises. However, the clinical signs, mortality and production losses were more severe in the older birds at Site A. Finally, the suspicion of AND was reported promptly by the PVS, enabling the rapid investigation and diagnosis of disease. The threat of incursion of avian influenza viruses remains, and vigilance for suspected clinical disease is advised.
Pigeon paramyxovirus type 1 (PPMV-1) investigations
In total, six submissions relating to the investigation of five cases of suspected PPMV-1 infection in lofts of pigeons were received during the quarter. Four of the investigated cases were located in England (Essex, Hampshire, Hertfordshire and Suffolk). One of these four cases resulted in the submission of serum samples only (five), and positive haemagglutination inhibition (HI) test titres (≥ 24) were recorded for 4/5 serum samples (HI titre range 24 – 28). Investigation of the other three suspected cases resulted in the submission of pigeon carcasses, and samples were subject to attempted virus isolation. No other haemagglutinating virus (including PPMV-1) was detected. The fifth case was located in the Scottish Borders and resulted in laboratory testing of samples from two submissions (five sera and samples from pigeon carcasses). Evidence of PPMV-1 infection was detected serologically (3/5 sera positive; HI titre range 24 - 27), and by virus isolation in embryonated fowls’ eggs. Serology was completed on all blood samples according to the standard protocol (CEC, 1992).
PPMV-1, a virulent variant ND virus, is the causative virus of the continuing panzootic that began in racing and feral pigeons almost 30 years ago. Although a pigeon variant virus, PPMV-1 still meets the internationally recognised virulence criteria, and under EU legislation when it is found in any poultry species the infection must be regarded as Newcastle disease.
Zoonoses
Salmonella
| In the tables and figures below, an incident is defined as ‘the first isolation and all subsequent isolations of the same serovar or serovar and phage/definitive type combination of a particular Salmonella from an animal, group of animals or their environment on a single premises, within a defined time period (usually 30 days). |
No clinical cases of disease due to S. Enteritidis have been recorded on VIDA in chickens during the quarter, or since 2004 when the last case was recorded.
Sampling of chicken layer flocks according to the requirements of the Salmonella National Control Programme (NCP) for layers is ongoing. More details on the Salmonella NCP in layers can be found on Defra’s web site.
The annual number of incidents of S. Enteritidis and S. Typhimurium in turkeys is shown in Table 5 below, and of S. Binza and S. Orion in pheasants in Table 6. In both of these tables the figures for 2009 (1st Quarter) are provisional.
| Table 5. The annual incidents of S. Enteritidis and S. Typhimurium in turkeys | |||||
| 2005 | 2006 | 2007 | 2008 | 2009 | |
|---|---|---|---|---|---|
| Enteritidis (total) |
0 | 0 | 0 | 0 | 0 |
| Typhimurium (total) |
23 | 38 | 12 | 1 | 0 |
Note: The incidents of S. Enteritidis and S. Typhimurium exclude isolates arising from the 2006/07 EU survey of turkey flocks (see Avian Quarterly Report, 10 (3), July-September 2006, Appendix 1).
| Table 6. The annual incidents of S. Binza and S. Orion in pheasants | |||||
| 2005 | 2006 | 2007 | 2008 | 2009 (Q1) | |
|---|---|---|---|---|---|
| Binza (total) |
10 | 21 | 7 | 6 | 0 |
| Orion (total) |
3 | 3 | 2 | 2 | 0 |
Cross-Sector Avian Diseases
Blackhead (histomonosis)
No incidents of blackhead were recorded on VIDA in chickens and turkeys during the quarter. However, there was an anecdotal report of an outbreak in a housed turkey breeder flock where the means of entry was considered likely to have been contaminated clothing or footwear.
Fowl Cholera (Pasteurella multocida)
No incidents of fowl cholera (Pasteurella multocida) were recorded on VIDA in chickens or turkeys during the quarter.
Marek’s Disease
The gradual decline in the number of incidents of Marek’s disease recorded on VIDA continued during the quarter (Figure 13). Among the incidents recorded in small backyard or hobby flocks was a case in a six-month-old light Sussex chicken, which showed a typical clinical presentation of leg paralysis on one side, and post-mortem examination confirmed gross enlargement of the sciatic nerve on the same side but there was also marked thickening of the caecal wall and tumour-like lesions in the lungs. Changes typical of Marek’s disease were demonstrated by histopathology in all three tissues. The combination of both nerve enlargement (the ‘classic’ form of the disease) and visceral tumours (the ‘acute’ form of the disease) was unusual, and the location of the lesions in the lungs and caecal wall was also unusual. The bird had not been vaccinated against Marek’s disease.
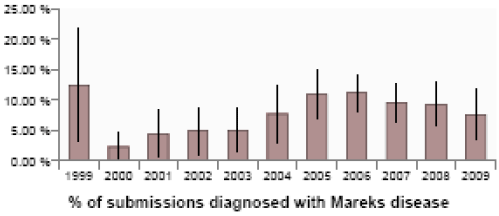
(Vertical bars represent 95 per cent confidence limits)
Endemic Disease Surveillance
Commercial Layers and Layer Breeders
Erysipelas
Further cases of erysipelas (Erysipelothrix rhusiopathiae) septicaemia causing prolonged moderately elevated mortality, and resulting in very high cumulative mortality, were seen in three flocks of organic free-range layers aged 40, 65 and 76 weeks in different parts of the country (see Quarterly Report October-December 2008, page 15). There is a clear case for vaccination of birds destined for premises with a history of this condition, and probably also as an aid to control in the face of an outbreak. There is currently a need for such a vaccine licensed for use in chickens in the UK.
Necrotic enteritis
An unusual case of necrotic enteritis was diagnosed in a flock of 10000, 29-week-old free-range layers with a history of sudden slight increase in mortality. Typical lesions were found in two of the three birds submitted for post-mortem examination and involved almost the entire intestinal tract, sparing a small section around Meckel’s diverticulum. Lesions were confirmed histologically as large areas of coagulative necrosis in which there were numerous colonies of large Gram positive bacilli. Necrotic enteritis in layers of this age is very unusual. In broilers there is sometimes an association between coccidial infection and necrotic enteritis. Coccidiosis is occasionally seen in layers in the early part of lay, although no evidence of it was found in these birds. No possible predisposing factors were discovered in this case.
This is an unusual manifestation of this condition. This fact together with the absence of the usual predisposing factors indicates that further monitoring is indicated should further cases occur.
Broilers and broiler breeders
Submissions of batches of broiler chickens for post mortem examination were slightly fewer than usual for this quarter than in recent years. Mostly they were of common conditions such as navel and yolk sac infection and E. coli infections including chronic polyserositis, abdominal cellulitis and septic arthritis. Two cases of suspected “spiking mortality” (probable hypoglycaemia) were seen in 25- and 28-day-old flocks and birds from two other flocks showing unevenness and stunting were examined (discussed in detail in Quarterly Report July-September 2008, page 11). There were also histopathology submissions from poultry practitioners from episodes of probable “spiking mortality”.
Chronic spondylitis
Enterococcus cecorum was isolated from spinal lesions in 41-day-old birds with chronic spondylitis (“spinal abscess”), a condition which appears to have declined markedly since it became very common for a period of a few years around six or seven years ago (Wood and others, 2002, Isolation of Enterococcus cecorum from bone lesions in broiler chickens, Veterinary Record, 150: 27).
Gangrenous dermatitis
A severe episode of gangrenous dermatitis, a condition now very uncommon in broiler chickens, was seen in February. Two houses of approximately 30,000 thirty-day-old birds showed a sudden onset of high mortality, with up to 1400 dead on two consecutive days in one house. The two affected houses were from a common source and two other unaffected houses on-site were of a different breed, and from different parent flocks in a completely different location. Classical lesions of gangrenous dermatitis were present in all birds examined and staphylococci and Clostridium septicum were isolated from lesions. This condition is thought to be commoner in immuno-suppressed birds, particularly those derived from parent flocks not vaccinated for chick anaemia virus (CAV) and which become infected once in lay. The CAV vaccinal status of the parents of the affected flocks was in doubt but could not be ascertained. Birds examined at post-mortem had chronic bursal atrophy. Animal Health was notified, but avian notifiable disease was excluded.
There were very few submission of broiler breeders this quarter. A further case of traumatic Achilles tendon rupture in females in the early part of lay was seen (see Quarterly Report October-December 2008, page 16). Enterococcus hirae-associated encephalomalacia was diagnosed in six-day-old parent stock chicks. This condition occurs between day 3 and day 8 and is not uncommon in broiler chicks, less so in layer pullets, and is unusual in broiler breeders.
Turkeys
Wet litter problem
An episode of “wet litter” occurred in a group of 5,000 seven-week-old turkey poults. Huddling and ruffled feathers were described and approximately 20 per cent of the birds were affected. Two birds submitted live had relatively little food in the crops and gizzards and large intestinal contents were described as undigested. Lesions of acute mild enteritis/typhlitis were non-specific. Rotavirus was detected in intestinal contents of one of the two birds. No significant bacteria were isolated from these two birds nor spirochaetes cultured anaerobically. Avian metapneumovirus has been associated with wet litter. In this case, sera from the two live birds was negative on ELISA testing for antibodies to this agent.
In the same quarter, rotavirus was confirmed in a faecal sample from a two-and-a-half-week old turkey poult. The clinical history indicated high mortality in young turkeys with vocalisation and severe typhlitis.
Ducks and Geese
Botulism was suspected, although not proven, in an incident of weakness and death described in ornamental ducks. Eight out of thirty ducks died in a group kept outside in a caged duck pond. Clinical signs consisted of a progressive weakness over a period of several days leading to death. Birds had difficulty walking and flying and wing drooping was described. Although there was no obvious source of botulinum toxin, it was suggested that carcasses of previously dead birds in the past may have sunk to the bottom of the pond.
Aeromonas hydrophila/caviae and Pseudomonas aeruginosa were isolated from the carcass of a goose which died suddenly. Gross lesions included perihepatitis, peritonitis and pericarditis with splenomegaly and the bacteria isolated were in septicaemic distribution. It was the only bird affected in a group of 30 birds. P.aeruginosa can be a pathogen of poultry (e.g. associated with yolk sac infections in hatcheries with contaminated water). It is generally thought to be opportunistic in nature. A.hydrophila has been isolated in both local and septicaemic infections in birds, particularly ducks.
Backyard Flocks
Concerns regarding biosecurity in the backyard flock sector were illustrated by the following disease outbreaks.
One hen from a group of 15 was presented for post-mortem examination with a three-day history of disorientation, anorexia, yellow diarrhoea, cyanosis of the comb and head twitching. All the remaining birds in the group were reported to have cyanosis of the comb and anorexia. There was a history on the premises of ‘rescue’ birds being introduced and there was recent contact with a premises where there were a number of chickens from different sources. Avian notifiable disease was considered a possibility and investigations were immediately made to rule this out. Egg peritonitis was found to be the cause of death in the bird presented.
On a second premises, a new cockerel was introduced to a small flock of 50 chickens and eight days later respiratory signs began to be seen in the group. Infectious laryngotracheitis (ILT) was confirmed in one of the birds that died. This disease, caused by herpes virus, is often seen after being introduced to susceptible flocks with the introduction of purchased birds. Purchased birds can appear healthy but may be carriers of the virus.
The owners were given appropriate advice about the risk of introducing infectious diseases.
Game Birds
There were very few game bird submissions in the quarter, as few disease problems are generally recognised during the over-wintering period, prior to the start of the breeding season. However, two diagnoses were of note. The first was a report of heavy infestations with the louse Menacanthus stramineus. This is the most pathogenic louse of poultry and can also occur on pheasants. Like other louse species, heavy infestations often occur concurrently with debilitating problems such as internal parasitism and poor nutrition, particularly during winter.
The second diagnosis of note was of the caecal worm Heterakis isolonche in an ornamental pheasant that had died. Although formerly well recognised in pheasants, H. isolonche is now rarely recorded, probably as a result of the widespread use of anthelminthics. It causes severe disease in the caecum and is much more pathogenic than the more widespread caecal worm, H. gallinarum, which parasitises a wider range of host species and is generally associated with few clinical signs. Although H. isolonche responds well to anthelminthic treatment in the bird, the eggs can remain viable in the soil for a year and there is therefore the risk of reinfection in birds using the same ground.
Further Reading
| - | You can view the full report by clicking here. |
Further Reading
| - | Find out more information on the diseases mentioned in this article by clicking here. |
June 2009











