



Better control of infectious bursal disease starts in the hatchery
Ceva innovates in India with the introduction of the world’s leading immune-complex IBD vaccine, Transmune IBD, to enable customers to vaccinate birds efficiently in the hatchery.Infectious bursal disease: a disease of socio-economic importance
Infectious Bursal Disease (IBD) is an acute, highly contagious viral infection in young chickens, predominantly affecting the immune system, with the bursa of Fabricius as the main target organ. The disease was first described by Cosgove in 1962 and the disease is now recorded in most parts of the world. The emergence of a “very virulent” form of IBD (vvIBD) has also been causing significant economic losses and IBD is considered a disease of socio-economic importance at the international level. The IBD virus replicates in the bursa of Fabricius and causes bursal inflammation and destruction of developing B lymphocytes. The IBDV infection can cause immunosuppression and a decrease in vaccine response, making chickens susceptible to secondary bacterial infection.
Prevalence of IBD in India
The presence of vvIBD in Indian poultry flocks has been recorded as early as 1993. In India, vvIBD strains have been implicated in several field outbreaks in recent years, despite routine vaccination. According to the official OIE database, IBD outbreaks have been occurring continuously since 2005. The number of outbreaks is presented annually in the following table.
|
Summary of IBD outbreaks reported in India based on OIE Database |
||||||||||||
|
2005 |
2006 |
2007 |
2008 |
2009 |
2010 |
2011 |
2012 |
2013 |
2014 |
2015 |
2016 |
2017 |
|
249 |
260 |
177 |
93 |
127 |
169 |
338 |
210 |
273 |
183 |
116 |
124 |
90 |
A number of factors such as poor vaccination practices, improper handling of the vaccine, breaks in cold chain during transport and at the farm, and the use of chlorinated water during vaccination are some potential factors that may contribute to the occurrence of disease in the vaccinated flocks. More importantly, the development of active immunisation in maternally immune chickens is impacted by the varied levels of maternally derived antibodies (MDA) in the chickens which affects the proper time of vaccination.
A local study conducted in 2018 screening about 1,600 serum samples collected from 86 farms highlighted that only 55 percent of the birds would have been vaccinated at the right time if the flocks had been vaccinated with live attenuated IBD vaccines at the ages of 6 and 12.
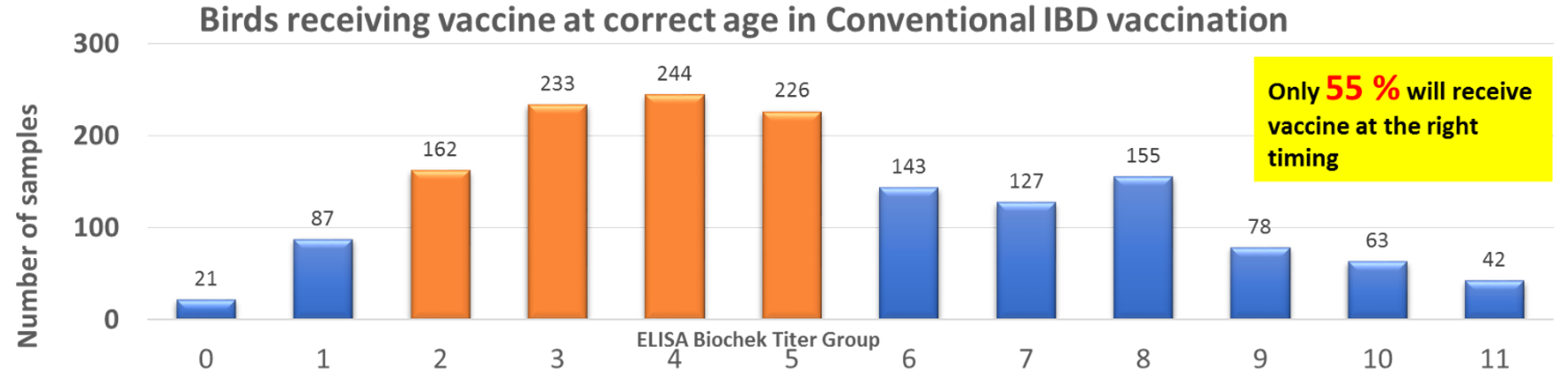
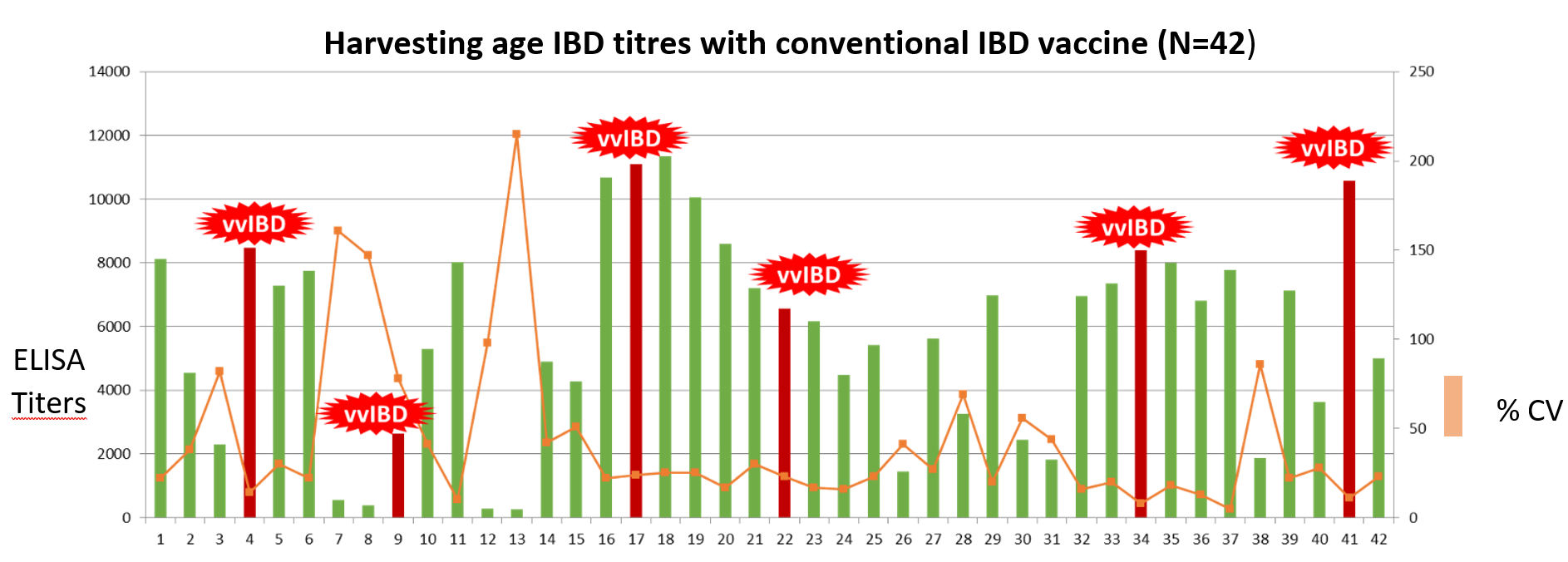
Moreover, the chickens vaccinated with the conventional farm vaccination programme showed huge variation of titres at harvesting age and about 15 percent of farms were detected with vvIBD.
Ceva brings innovative solutions to India for better IBD control
Ceva addressed the above problems of inaccurate vaccination and interference with maternally derived antibodies by introducing IBD vaccination in the hatchery with its innovative vaccine, Transmune IBD, along with vaccine delivery systems well supported by its hatchery vaccination team. Transmune IBD is an innovative immune-complex IBD vaccine, prepared from live attenuated Intermediate Plus IBD (strain Winterfield 2512) mixed together with specific anti-IBDV serum (Virus Protecting Immunoglobulins, VPI) in a well formulated proportion. The vaccine virus is shielded and is not recognised or neutralised by the MDA. The VPI catabolises in the body in same way as MDA, the free vaccine virus is released from the immune complex and break through the MDA at the optimal time and colonise the bursa. The time of the release and the onset of replication in the bursa are dependent on the level of MDA; the lower the MDA the sooner the release and replication of the vaccine virus. With administration in the hatchery using proper equipment, every chicken receives an accurate dose of the vaccine.
Globally, the poultry producers are shifting towards hatchery vaccination and today almost 45 percent of broilers produced in the world are vaccinated against IBD in the hatchery using new technology vaccines like Transmune IBD. The poultry industry in India has also undergone a major shift in recent decades with integrated broiler producers contributing around 85 percent of total broiler production. The broiler segment is growing at around 8-10 percent annually and broiler producers are looking for innovative and more efficient animal health solutions, including vaccines.
Experience using Transmune IBD in India
Ceva, being committed to innovation in the hatchery, has always been a leader in hatchery vaccination solutions, equipment and services. During its introduction in India, Ceva offered key broiler producers with a complete vaccination solution involving new technology vaccines, vaccination equipment, services and monitoring programmes for customers to realise the full value of their vaccination solutions. All these form a part of the Ceva Innovation Areas like C.H.I.C.K. Program (Ceva Hatchery Immunization Control Keys), Global Protection Services (GPS) and Lab services, among others.
Since its launch in India during Q2 2018, Transmune IBD has been rapidly adopted by many key integrators in their broiler vaccination programmes and tens of millions of birds were vaccinated with this innovative vaccine each month. The vaccination programme is well monitored through the Ceva C.H.I.C.K. Program and the VSE Team (Vaccination Services and Equipment). The C.H.I.C.K. Program involves Ceva VSE specialists visiting producers’ hatcheries at regular intervals and auditing the quality of vaccination. The aim of this programme is to ensure that all birds are well vaccinated and to train the vaccination teams to achieve the highest quality of hatchery vaccination.
At the farm level, the vaccinated flocks are monitored through a disease monitoring programme called Global Protection Services (GPS). GPS allows for the monitoring of flock health over time and measures the vaccine performance of Transmune-vaccinated flocks. Since its introduction in India, over 2,500 serum samples have been monitored.
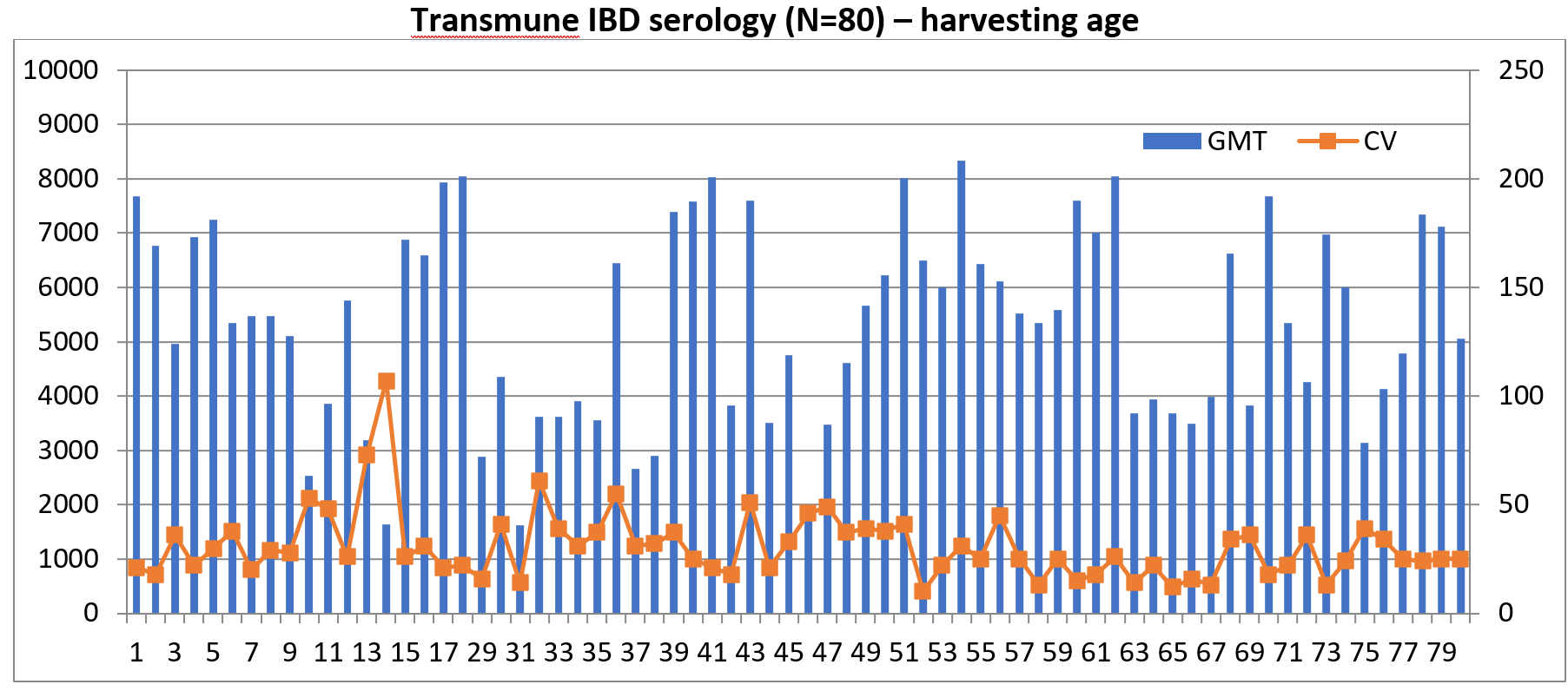
Vaccination with Transmune shows development of more uniform titres and better flock coverage leading to better flock protection against IBD. Several field trials have been conducted in comparison to the conventional IBD vaccination programme showing better performance and value in terms of FCR, liveability and body weight gain in Transmune-vaccinated flocks compared to the other group.
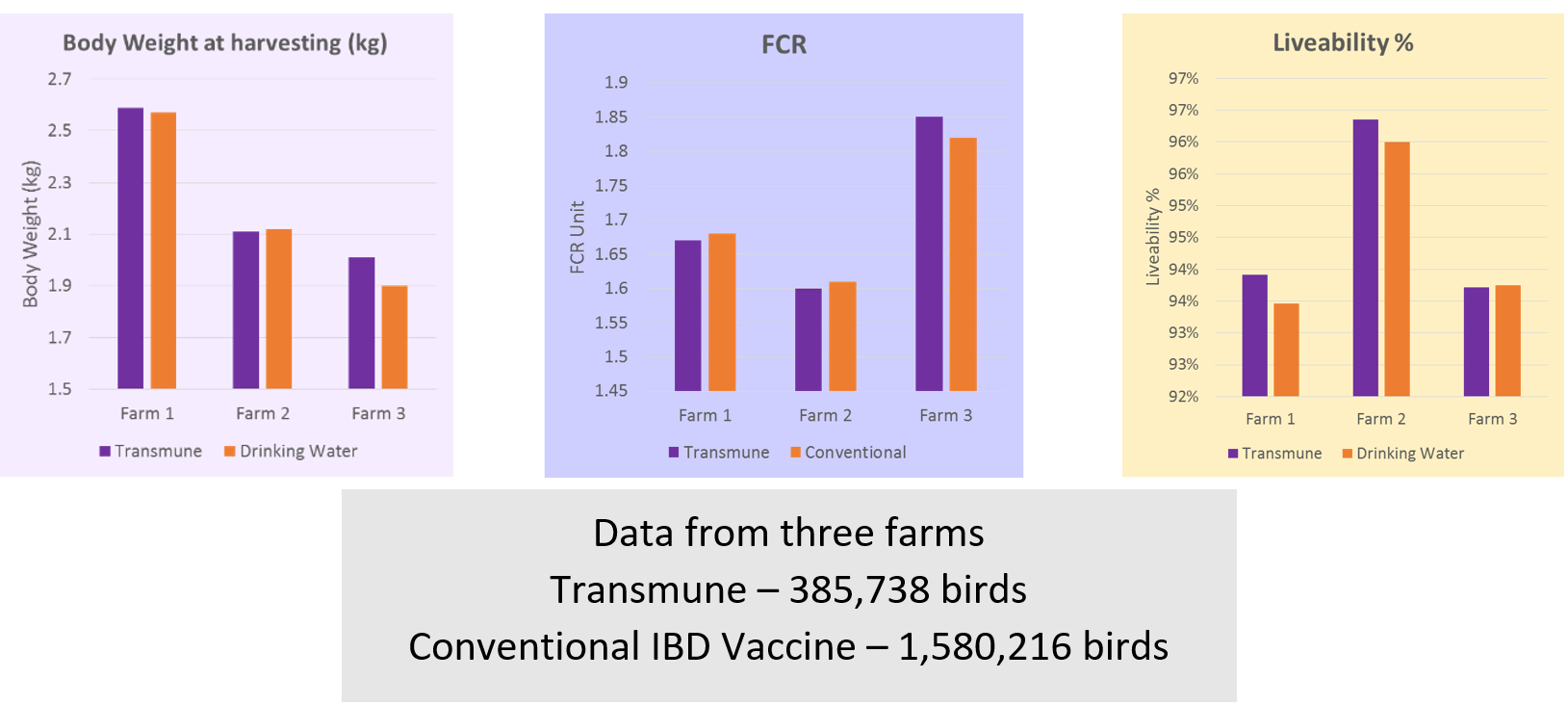
These trials demonstrated the improvement in performance at the farm level in Transmune-vaccinated flocks with better control over clinical and sub-clinical forms of IBD. By blocking the bursa to field the IBD virus infection, Transmune IBD stands out with its unique ability to stop the cycle of replication and spread of the virus. Sub-optimal control over IBD challenges can reduce flock performance, increase antibiotic usage in the farm, lead to immune-suppression and increases in other secondary infections.
India continues to be one of the fastest growing poultry markets and Ceva will continue to introduce new innovative concepts to the Indian market like new technology vaccines plus automated equipment to enable a shift to vaccination at the hatchery and continuously improve the quality of vaccination. At the next stage, Ceva in India shall apply for the first ever Quality Recognition of its hatchery vaccination service through a third-party auditing company and continue to invest and innovate at the hatchery level and day-old chick health.










