Effects of three different live vaccination schemes against infectious bronchitis on the performance of broilers under field conditions
Avian infectious bronchitis (IB) is a major economic burden to poultry and, though its control is mainly based on vaccination with one or more serotypes of Avian infectious bronchitis virus (IBV), the effects of different vaccines on the performance of broilers under field conditions was seldom assessed.written by Lucas Pedroso Colvero1, Thiago Moreira Tejkowski1, Paulo Eduardo Brandão, Sueli Akemi Taniwaki2 and Laura Yaneth Villarreal, MSD Animal Health Brazil. Avenida Dr. Chucri Zaidan. São Paulo, SP, Brazil, Department of Preventive Veterinary Medicine and Animal Health, School of Veterinary Medicine, University of São Paulo, Av. Professor Dr. Orlando, Marques de Paiva, São Paulo, SP, Brazil.
Abstract
Day-old broilers were spray-vaccinated with live-IBV vaccines containing strains Ma5 (Group A), H120 (Group B) or H120 plus BR-I (Group C), with 49,000 birds/ group and kept under field conditions. IBV typing revealed that no IBVs other than GI-11 (Brazil type) were present on Groups A, B and C throughout the experiment, with a high nucleotide identity with the BR-I vaccine strain.
Tubular dilatation and epithelial shortening with discrete to moderate foci of degeneration and perilesional hemorrhage were found in kidneys of group C. As Group A showed the higher performance parameters (lower death ratio and higher daily weight gain and weight at slaughter), relative total economic loss/ 49,000 broilers for groups B/ C were US$ 522.88/ US$ 82.39, respectively, showing that broilers vaccinated with Ma5 could in some situations outperform those vaccinated with H120 or with the combination of H120 plus BR-I from the performance parameters and economic point of views.
Introduction
Amongst the diverse causes of loss of profitability in poultry, including management, infectious and nutritional diseases and susceptibility to market fluctuations, avian infectious bronchitis (IB), a worldwide spread endemic infectious disease, is a chief source of economic losses in broilers and breeders [1; 2].
IB control is focused on the use of live (in broilers) and live plus inactivated (layers and breeders) vaccines with either a single strain or the combination of two strains of the causative agent (Avian infectious bronchitis virus IBV, a Gammacoronavirus).
Dual vaccination with some serotipically distinct live strains is known to broaden the protection spectrum against IBV [3] due do a phenomenon known as protectotype. Nonetheless, the cross-protection amongst the many different serotypes of IBV described hitherto might be low and not predictable by variations on the neutralising antibodies-inducing spike glycoprotein [4], making vaccine selection an experimental-challenge or even field-based procedure.
Until 2016, Massachusetts-type strains (GI-1), including Ma5 and H120, were the only ones allowed by the Brazilian Ministry of Agriculture to be used as live vaccines but on 2017 a Brazil-type (GI-11) live vaccine based on a strain named BR-I has been released in this country.
If IB is not properly controlled, the performance of birds might be affected by the development of respiratory diseases, enteritis, nephritis, infertility and egg drop [5-8], but quantitative data on the effects of different vaccination strategies on the performance of broilers to provide rationale for producers when it comes to IB vaccination is lacking.
This investigation was designed to compare the effects of live IBV vaccines on the pre-slaughter performance of broiler flocks using a model based on live Ma5, H120 or BR-I plus H120 live IBV vaccines under field conditions in Brazil and in association with pathological and antibody responses bases.
Materials and Methods
Birds. A total of 147,000 Cobb500 day-old broilers were included in this study and divided in three groups/ flocks (A, B and C) with 49,000 birds/ group/flock.
All groups were kept simultaneously but in separate buildings on a same commercial broiler farm in Southeastern Brazil until slaughter (46 days of age) and were housed and fed as per Cobb’s manual.
Experimental design. All birds were vaccinated at a day old via spray at hatchery with commercially available live vaccines against IBV containing strains Ma5 (Group A), H120 (group B) or BR-I plus H120 (group C); IBV strains in each vaccine had a minimum of 103.0 DIE50/dose. At the same moment, all birds were also vaccinated against IBDV and NDV.
Groups and vaccination schedules were under a double-blind protocol, i.e., the farmer and the authors were not aware of which flock received which vaccine. All vaccination protocols and doses were as per manufacturer’s instructions and were chosen based on the protocols currently used in Brazil in commercial broiler farms.
Sera were collected from 12 randomly selected birds/group at weeks 2, 4 and 5 of age and at slaughter and tested for anti IBV antibodies (dilution=1:500) with the generic IDEXX® IBV Ab Test ELISA (IDEXX Laboratories). ELISA titers were compared within each group and amongst all groups along the four sampling times with the Kruskal-Wallis test at a significance level of 0.05 using SOCS at http://www.socscistatistics.com.
Samples of trachea plus lungs (=respiratory samples), kidneys and cecal tonsils were also collected from other randomly selected birds in FTA® cards (Whatman) from 5 birds per group at weeks 1, 2, 4 and 5 of age and at slaughter and tested for IBV RNA using an RT-PCR targeted to the 3’UTR of the virus genome as described by Cavanagh et al. [9]. Briefly, RNA of each sample was extracted from ¼ of each FTA card circle with PureLink™ RNA mini kit (Thermo Fisher) and submitted to reverse transcription with random primers and M-MLV Reverse Transcriptase (Invitrogen), following manufacturer’s instructions. Amplification was carried out in a total volume of 25 µL reaction containing 1 × GoTaq® Green Master Mix (Promega), 400 nM of each primer UTR41+ and UTR11- (PCR) or UTR41+ and UTR31- (hemi nested-PCR) and 2.5 of RNA or 1 µL of DNA product, respectively to PCR and hemi-nested PCR. Amplification conditions were: 94 ºC/4 min.; 35 cycles of 94 ºC/15 s, 48 ºC/15 s and 72 ºC/30 s; and 72 ºC/10 min., and amplicons of the expected size of 179 bp were visualized by 1.5% agarose gel electrophoresis stained with SYBR™ safe DNA Gel Stain (Invitrogen) in comparison with GeneRuler 100 bp DNA ladder (Thermo Fisher).
Positive samples were submitted to quantitative RT-PCR (RT-qPCR) using Power SYBR® Green RNA-to-Ct™ 1-Step kit (Applied Biosystems), 250 nM of primers IBV5’GU391 and IBV5’GL533 [10] and 2 µL of RNA samples in triplicate, following manufacturer’s instructions, for viral genomic RNA measurements. Chicken β-actin RT-qPCR for all RNA samples was used as endogenous control with Power SYBR® Green RNA-to-Ct™ 1-Step kit (Applied Biosystems), 400 nM of primers Chick β-actin F and R [11] and 2 µL of RNA in duplicate, following manufacturer’s instructions. Both RT-qPCRs were carried out in 7500 Real-Time PCR system (Applied Biosystems) with same cycling conditions (48 ºC/30 min; 95 ºC/10 min; 45 cycles of 95 ºC/15 s and 60 ºC/40 s; and melting curve analysis), but in separated tubes. Absolute quantification (number of copies/ µL of RNA) of IBV was made in comparison with ten-fold dilution standard curve ranging from 1 × 103 to 1 × 109 copies per reaction (slope=-3.536 and y-intercept=43.665) normalized with chicken β-actin Ct value of each sample.
IBV typing based on partial S1 sequencing was performed in positive samples, using hemi-nested PCR as described by Torres et al. [12]. Briefly, reaction was carried out in a total volume of 25 µL reaction containing 1 × GoTaq® Green Master Mix (Promega), 400 nM of each primer Braco F and Braco R (PCR) or Braco F and Braco R2 (hemi nested-PCR), and 2.5 of RNA or 1 µL of DNA, respectively to PCR and hemi-nested PCR. Amplification conditions were: 94 ºC/3 min; 40 cycles of 94 ºC/15 s, 50 ºC/30 s and 72 ºC/45 s; and 72 ºC/10 min, and amplicons of expected size of 450 bp were visualised by 1.5% agarose gel electrophoresis stained with SYBR™ safe DNA Gel Stain (Invitrogen) in comparison with GeneRuler 100 bp DNA ladder (Thermo Scientific). Positive fragments were excised from gel, purified with Illustra GFX PCR DNA and Gel Band Purification kit (GE Healthcare), following manufacturer’s instructions, and submitted to bidirectional sequencing with BigDye™ Terminator v.3.1 Cycle Sequencing kit and 3500 Genetic Analyzer (Applied Biosystems).
All samples negative to the3’UTR were tested by the endogenous control qRT-PCR in order to check for false negatives at this step.
At week five, tissue samples from trachea and kidneys were collected from 5 randomly selected birds per group, fixed in 10% formalin and processed for histopathological examination with hematoxylin and eosin (HE) staining.
Performance parameters. The groups were monitored for the Daily Weight Gain (DWA), Efficiency Index (EI=DWG x viability /feed conversion ratio x 100) and weekly and accumulated death-plus-discarded ratio, i.e., the ratios of birds found dead or that were discarded from the flock due to disease or other reasons regarding the total population/flock.
Economic estimates for the three flocks were based on US$ 0.93/kg of broiler meat at slaughter taking into account the death-plus-discarded ratios and weight at slaughter considering the best performance group as a reference
Results and Discussion
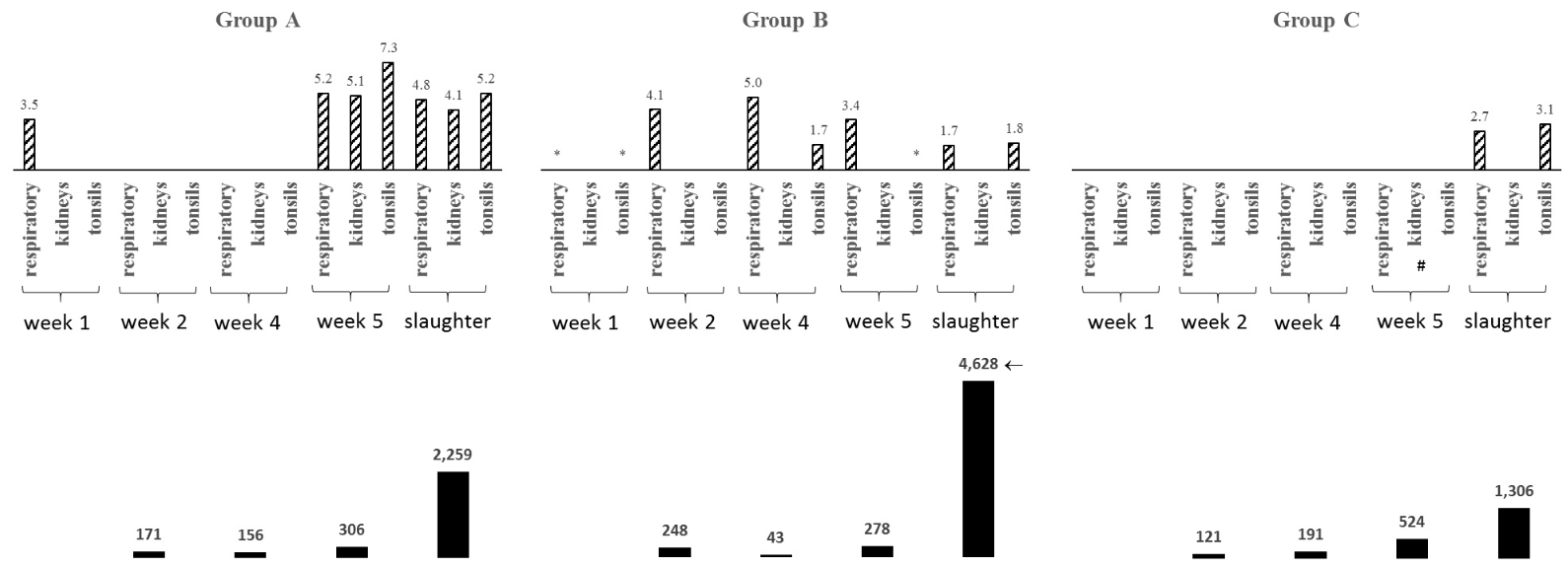
RT-PCR for IBV RNA, ELISA for anti-IBV Abs detection and histopathology. IBV RNA was found in week 1 for both groups A (respiratory samples, 3.5 log copies/µL) and B (respiratory samples and cecal tonsils, qRT-PCR results bellow detection level). All samples from group A were PCR-negative at weeks 2 and 4 and positive at week 5 and at slaughter, with viral loads on respiratory tract, kidneys and tonsils of 5.2, 5.1 and 7.3 and 4.8, 4.1 and 5.2 log copies/µL for each of these two sampling times, respectively (Fig. 1).
Fig. 1. IBV viral load (log copy number/RNA microliter) in broilers vaccinated with live Ma5 (Group A), H120 (Group B) or BR-I plus H120 (Group C) IBV strains at day old, from week 1 to slaughter, using qRT-PCR in samples of the respiratory tract, kidneys and cecal tonsils (upper graphs) and anti-IBV ELISA Abs mean titers from week 2 to slaughter (lower graphs). Samples negative to qRT-PCR but positive to conventional RT-PCR are indicated with *; #=moment at which histopathological lesions were found (only in kidneys of Group C at week 5); the arrow indicates amongst-groups significant difference. All IBVs were typed as GI-11 (Brazil) type.
For group B, from week 1 onwards, kidneys were negative for IBV RNA, while, on respiratory tract, IBV RNA was found in all sampling times, with viral loads peaking at week 4 (5.0 log copies/µL), while tonsils viral loads peaked at slaughter (1.8 log copies/µL) (Fig. 1).
No IBV RNA was found for group C from weeks 1 to 5 and only at slaughter positive PCRs were found for respiratory tract and tonsils (2.7 and 3.1 log copies/µL, respectively), while kidneys showed negative results (Fig. 1).
All samples negative to the3’UTR were positive for the chicken beta-actin endogenous control qRT-PCR with Cts ranging from 15.41 to 24.04.
Fig. 2. Nucleotide alignments for partial S1 sequences (positions 20,566 to 20,716 regarding BR-I vaccine strain Genbank Accession # KY626044) for IBV strains detected in broilers vaccinated with live Ma5 (Group A), H120 (Group B) or BR-I plus H120 (Group C) IBV strains at day old and the respective sample types and week of detection.
IBV typing (successful for 13 of the IBVs delectated, Fig. 2) revealed that no IBV types other than GI-11 (Brazil type) were present on Groups A, B and C throughout the experiment, with S1 nucleotides identities ranging from 90-91% (group A and B, respiratory sample, weeks 1 and 4, respectively) to 99 % (all other IBV positive samples) regarding the BR-I vaccine strain used in this study (Genbank accession number KY626044). As the sequences were < 200nucleotides, they were not submitted to the Genbank, but are available upon request; a nucleotide alignment is shown in Fig. 2 for the sequences that were obtained.
Ameans for group C showed an increasing pattern from week 2 to slaughter while, for groups A and B, Ameans decreased from week 2 to 4 and increased from week 4 to slaughter (Fig. 1).
As seem in Fig. 1, the lowest Amean at slaughter was found for Group C (1,306), while, for Groups A and B Ameans at slaughter were 2,259 and 4,628, respectively.
Comparing ELISA Ameans for anti-IBV antibodies between groups at each sampling time (shown in Fig. 1), only for Group B at slaughter the titers were significantly higher, with p values of 0.02133 and 0.00072 and when compared to Groups A and C, respectively. All within-group ELISA titers showed a significant variation along the four sampling times.
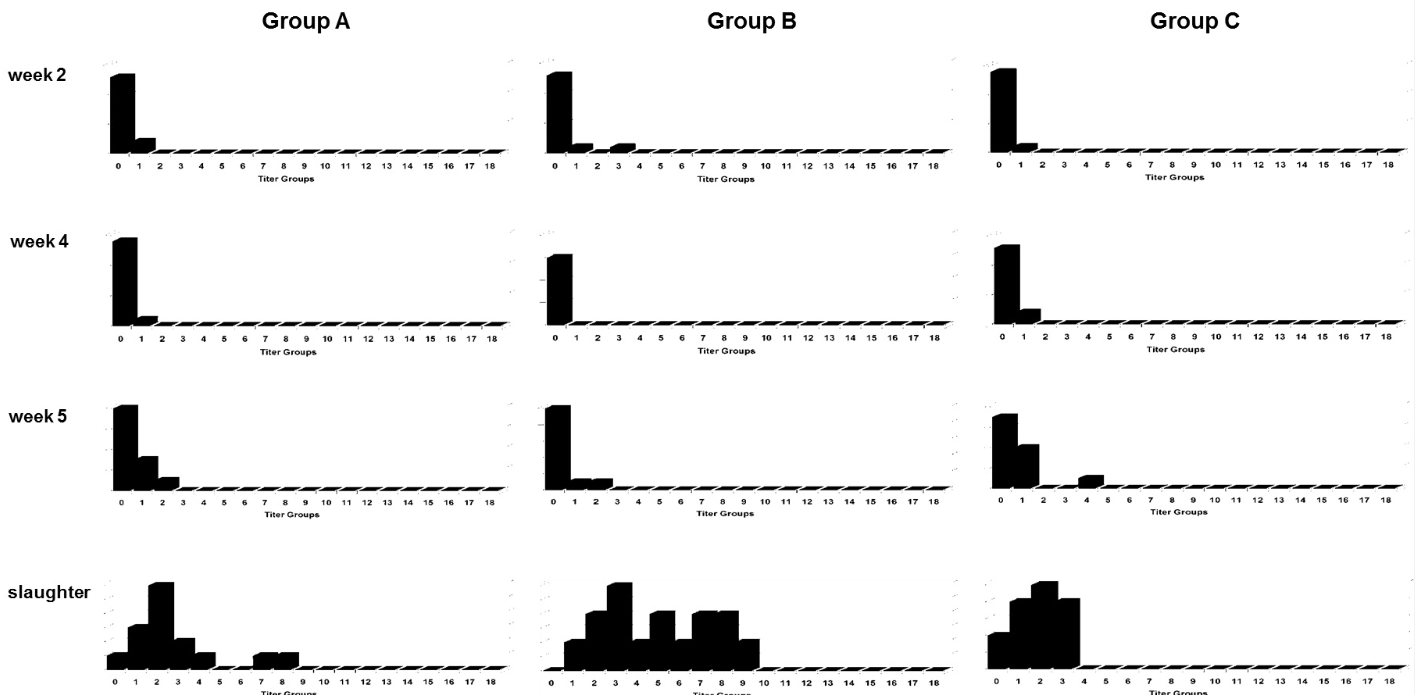
Group B (vaccinated with H120) showed the highest heterogeneity of anti-IBV Abs titers at slaughter (Fig. 3), with no evident difference for the three groups at weeks 2, 4 and 5.
Fig. 3. Distribution of ELISA anti-IBV antibodies titers groups in birds from Groups A, B and C from week 2 to slaughter, vaccinated with different IBV live vaccines at day old. The Y axis represents the number of birds and the X axis the titer groups.
Histopathological examination of samples of tracheas at week 5 of age showed, for all three groups, a discrete to moderate lymphocytic infiltration and edema and degeneration of the mucosa.
At the same age, for Groups A and B, no alterations were found in kidneys; on the other hand, kidneys from birds from Group C showed a marked tubular dilatation and epithelial shortening with discrete to moderate foci of degeneration and perilesional hemorrhage.
Performance parameters. The accumulated death ratios before slaughter for groups A, B and C were 1.58, 2.85 and 1.76, respectively. Daily weight gain and weight at slaughter (in grams) for Group A were 60.79/2,736 and 60.43/2,719 for both groups B and C.
Finally, the efficiency index for groups A, B and C were 324, 319 and 322, respectively. As Group A showed the higher performance parameters (lower death ratio and higher DWG and weigh at slaughter), the relative total economic loss/ 49,000 broilers found for groups B and C were US$ 522.88 and US$ 82.39, respectively.
Cecal tonsils are known as a preferential site for IBV persistent infection [13] and this might be the reason for the IBV loads in all groups at this site. It’s noteworthy though that, for group A, cecal tonsils load decreased from week 5 to slaughter and peaked for groups B and C at slaughter, what could indicate a better protection from a carrier state in this group.
Regarding the respiratory samples, as shown in Fig. 1, group A remained clear of IBV at weeks 2 and 4 after being positive at week 1, while groups B was positive for all weeks with varying viral loads and C remained clear of IBV up to slaughter. All viruses detected in these respiratory samples showed S1 nucleotide identities of 99% with the BR-I vaccine strain used in group C, except for group A and B samples at weeks 1 and 4, respectively.
Live IBV vaccines are known to show a “rolling” effect, meaning that the vaccine can be transmitted from vaccinated to non-vaccinated birds [14]; taking this into account and the time lag for viral load increase along the experimental weeks, an identity of 99% in S1 with the vaccine strain BR-I for IBVs found in groups A and B might be considered as an indication of vaccine rolling from group C; however, it must be considered, that, due to the diversity of IBV lineages in Brazil [15], these S1s could still represent a field strain of GI-11 and not the vaccine itself.
Another indication that vaccine rolling might have played a role on IBV transmission is that two samples (group A and B, respiratory samples, weeks 1 and 4) contained IBVs that were diverse from the BR-I vaccine strain (90 and 91% nucleotides identities, respectively), what could be due to the known fast evolution of live IBV vaccine stains upon vaccination [14; 16]. On the other hand, the presence of a subpopulation of one of the four sub lineages of GI-11 (Brazil) type viruses found on the field in Brazil [17] in these two cases cannot be completely ruled out as an alternative explanation to vaccine rolling.
Taking into account that some IBVs in the GI-11 lineage have been described as being nephropathogenic [17], in the case of group C (vaccinated with the BR-I strain of the GI-11 lineage) it’s reasonable to argue that an homogeneous spray vaccination at a day old lead to a widespread protection against the colonisation of further organs during most of the experiment while, for group A, colonisation by BR-I via vaccine roll could have occurred at a later stage and in a non-homogeneous way, resulting in all samples being positive for this virus at week 5 and at slaughter.
When groups A and B are compared, anti-IBV Abs Ameans in B were more than 2-fold those of A at slaughter (p= 0.002) and the presence of BR-I like-viruses was widespread in all weeks (Fig.1). This could be primarily attributed to the higher antigenicity indexes in Ma5 S1 receptor-binding domain [18], what could result in a more effective protection from either vaccine roll or field challenge leading to lower field challenge-derived antibody titers.
Regarding the anti-IBV Abs titers groups (Fig. 3), for both groups A and B at slaughter a more heterogeneous distribution is noticeable, more prominently in B, when compared to C; heterogenous ELISA Abs groups are an indication of challenge instead of a vaccine effect and support, in this case, the aforementioned BR-I rolling hypothesis from C to A and B due to the absence of non-BR-I like viruses in these birds at slaughter.
The nephropathogenic potential of GI-11 IBVs is illustrated by the histopathological patterns found in kidneys of group C (vaccinated with BR-I) at week 5, including tubular dilatation, epithelial shortening and degeneration and perilesional hemorrhage, an indication of nephrosis. This histopathological pattern is largely compatible with previous report on nephropathogenic IBVs infection [3], including those on the GI-11 lineages in which BR-I is classified [17], suggesting either vaccine or field-induced kidney lesions.
Though IBV was not found in kidney samples in Group C in any sampling time, it must be mentioned that the pathogenesis of IBV in kidneys might take place not only under viral replication in these organs but also “at distance” in an immune mediated fashion without the presence of IBV at this site [19].
In a bigger picture, both the kidney disease in group C and the lower protection in group B might have accounted for the poorest performance of these groups when compared to group A, vaccinated with Ma5 both in terms of weight gain and death ratios.
Though the protectotype effect is well documented [3], not all combinations of IBV strains lead to renal protection even if respiratory protection is found [20]. This is in agreement with the findings in group C, in which a dual H120 plus BR-I vaccination was attempted but, despite the respiratory tract was protected, kidney lesions were still found, an indication that this combination of IBV strain might not be an efficient choice under field conditions.
Different types of IBV might interfere with the replication of each other in dual-vaccinated birds, as well as with different NDV vaccine strains based on competition by cell receptors [21; 22]. This heterologous interference might be speculated as having had some effect on the antibody levels and protection during this filed trial. Nonetheless, the interaction between the IBV types used herein amongst each other and with NDV has not been specifically assessed hitherto and in vitro assays would greatly improve the knowledge on this matter from the cell receptor competition point of view.
The impact of IBV on the broiler industry has been already described as substantial when the GI-11 (Brazil) lineage is involved [1]; to such a low-profit margin industry, an economic loss as that described for groups B and C (US$ 522.88 and US$ 82.39 per 49,000 broilers, respectively) is significant and shows how a proper vaccination program could help to lessen the impact of IB on broiler production.
These arguments are based on the explicit experimental conditions and laboratory procedures described along this manuscript based on the best available literature on this field but must be examined carefully though on the view of some practical experimental biases inherent to a field trial such as the one reported herein. Firstly, the role of vaccine roll should be investigated in a more elaborated manner under a controlled environment such as isolators to measure the time and mode of any vaccine spread. Second, full genome sequence for the IBV strains detected in the birds could shed light on their genealogy with respect to vaccine and field strains. Finally, regarding the performance parameters, only the bulk of the data was available instead of per bird data due to the high number (29,000) of birds per group; though this is the modus operandi of a real broiler farm, it does not allow the calculation of standard deviations or in-depth statistics amongst the groups.
Conclusions
Broilers vaccinated with Ma5 outperformed those vaccinated with H120 or with the combination of H120 plus BR-I from the performance parameters and economic point of views as a result of the absence of vaccine-induced histopathological changes. The procedures described herein can be applied to provided economic data for the choice of vaccination schemes on poultry.
References
[1] L. P. Colvero, L. Y. Villarreal, C. A. Torres, and P. E. Brandão, “Assessing the economic burden of avian infectious bronchitis on poultry farms in Brazil”, Revue Scientifique et Technique (International Office of Epizootics, vol. 34, no. 3, pp. 993-999, 2015.
[2] M. W. Jackwood, “ Review of infectious bronchitis virus around the world”, Avian Diseases, vol. 56, no. 4, pp. 634-641, 2012.
[3] M. Smialek, B. Tykalowski, D. Dziewulska, T. Stenzel, and A. Koncicki, “Immunological aspects of the efficiency of protectotype vaccination strategy against chicken infectious bronchitis”, BMC Veterinary Research, vol. 13, no. 1, pp. 44, 2017.
[4] J. J. de Wit, J. K. Cook, and H. M. van der Heijden, “ Infectious bronchitis virus variants: a review of the history, current situation and control measures”, Avian Pathology, vol. 40, no. 3, pp. 223-235, 2011.
[5] J. K. Cook, M. Jackwood, and R. C. Jones, “The long view: 40 years of infectious bronchitis research”, Avian Pathology, vol. 41, no. 3, pp. 239-250, 2012.
[6] R. C. Jones, “Viral respiratory diseases (ILT, aMPV infections, IB): are they ever under control?”, British Poultry Science, vol. 51, no 1, p.1-11, 2010.
[7] L. Y. Villarreal, P. E. Brandão, J. L. Chacón, M. S. Assayag, P. C. Maiorka, P. Raffi, A. B. Saidenberg, R. C. Jones, and A. J. Ferreira, “Orchitis in roosters with reduced fertility associated with avian infectious bronchitis virus and avian metapneumovirus infections”, Avian Diseases, vol. 51, no. 4, pp. 900-904, 2007.
[8] L. Y. Villarreal, P. E. Brandão, J. L. Chacón, A. B. Saidenberg, M. S. Assayag, R. C. Jones, and A. J. Ferreira, “ Molecular characterization of infectious bronchitis virus strains isolated from the enteric contents of Brazilian laying hens and broilers”, Avian Diseases, vol. 51, no. 4, pp. 974-978, 2007.
[9] D. Cavanagh, K. Mawditt, D. de B. Welchman, P. Britton, and R. E. Gough., “Coronaviruses from pheasants (Phasianus colchicus) are genetically closely related to coronaviruses of domestic fowl (Infectious Bronchitis Virus) and turkeys”, Avian Pathology, vol.31, no. 1, pp. 81-93, 2002.
[10] S. A. Callison, D. A. Hilt, T. O. Boynton, B. F. Sample, R. Robison, D. E. Swayne, and M.W. Jackwood, “Development and evaluation of a real-time Taqman RT-PCR assay for the detection of infectious bronchitis virus from infected chickens”, Journal of Virological Methods, vol. 138, no. 1-2, pp. 60-65, 2012.
[11] R. Yin, Z. Ding, X. Liu, L. Mu, Y. Cong., and T. Stoeger, “Inhibition of Newcastle disease virus replication by RNA interference targeting the matrix protein gene in chicken embryo fibroblasts”, Journal of Virological Methods, vol. 167, no. 1, pp. 107-111, 2010.
[12] C. A. Torres, A. S. Hora, P. O. Tonietti, S. A. Taniwaki, M. Cecchinato, L. Y. Villarreal, and P. E. Brandão, “ Gammacoronavirus and Deltacoronavirus in quail”, Avian Diseases, vol. 60, no. 3, pp. 656-661, 2016.
[13] J J de Wit, “Detection of infectious bronchitis virus”, Avian Pathology, vol. 29, no. 2, pp. 71-93, 2000.
[14] E. T. McKinley, D. A. Hilt, and M. W. Jackwood, “ Avian coronavirus infectious bronchitis attenuated live vaccines undergo selection of subpopulations and mutations following vaccination”, Vaccine, vol. 26, no. 10, pp. 1274-1284, 2008.
[15] L. Y. Villarreal, T. L. Sandri, S. P. Souza, L. J. Richtzenhain, J. J. de Wit, and P. E. Brandão, “ Molecular epidemiology of avian infectious bronchitis in Brazil from 2007 to 2008 in breeders, broilers, and layers”, Avian Diseases, vol. 54, no. 2, pp. 894-898, 2010.
[16] R. A. Gallardo, V. L. van Santen, and H. Toro, “ Host intraspatial selection of infectious bronchitis virus populations”, Avian Diseases, vol. 54, no. 2, pp. 807-813, 2010.
[17] J. J. de Wit, P. Brandão, C. A. Torres, R. Koopman, and L.Y. Villarreal, “Increased level of protection of respiratory tract and kidney by combining different infectious bronchitis virus vaccines against challenge with nephropathogenic Brazilian genotype subcluster 4 strains”, Avian Pathology, vol. 44, no. 5, pp. 352-357, 2015.
[18] C. L. M. Leyson, B. J. Jordan, and M. W. Jackwood, “ Insights from molecular structure predictions of the infectious bronchitis virus S1 spike glycoprotein”, Infection, Genetics and Evolution, vol. 46, pp. 124-129, 2016.
[19] C. W. Lee, C. Brown, D. A. Hilt, and M. W. Jackwood, “Nephropathogenesis of chickens experimentally infected with various strains of infectious bronchitis virus”, The Journal of Veterinary Medical Science, vol. 66, no. 7, pp. 835-840, 2004.
[20] J. Gelb Jr., B. S. Ladman, C. R. Pope, J. M. Ruano, E.M. Brannick, D.A. Bautista, C. M. Coughlin, and L. A. Preskenis, “Characterization of nephropathogenic infectious bronchitis virus DMV/1639/11 recovered from Delmarva broiler chickens in 2011”, Avian Diseases, vol. 57, no. 1, pp. 65-70, 2013.
[21] E. N. Ndegwa, S. N. Bartlett, H. Toro, K. S. Joiner, and V. L. van Santen, ”Combined infectious bronchitis virus Arkansas and Massachusetts serotype vaccination suppresses replication of Arkansas vaccine virus”, Avian Pathology, vol. 44, no. 5, pp. 408-420, 2015.
[22] J. Gelb Jr, B. S. Ladman, M. J. Licata, M. H. Shapiro, and L. R. Campion, “Evaluating viral interference between infectious bronchitis virus and Newcastle disease virus vaccine strains using quantitative reverse transcription-polymerase chain reaction”, Avian Diseases, vol. 51, no. 4, pp. 924-934, 2007.