



Predisposing Factors that Affect Walking Ability in Turkeys and Broilers
Edgar O. Oviedo-Rondón of North Carolina State University reviewed this topic at the 2007 Carolina Poultry Nutrition Conference.Importance of Gait from Nutritional and Production Perspectives
Walking ability in poultry is considered an important welfare parameter (Mench, 2004), but it is also critical for obtaining optimum flock performance, good efficiency of energy and feed utilization, reducing bone disorders, improving bone strength, reducing skeletal fractures, improving food safety and reducing processing plant condemnation issues.
It has been estimated that 2-6% of all turkey flocks and 1 or 2% of all broiler flocks display some observable signs of leg problems, while many more are affected in less visible ways. Gait evaluations in meat-type poultry flocks normally find between 30 to 65 % of the population with gait patterns called “abnormal” without having bone issues. Leg health is not always linked to bone disorders. The skeletal system is constituted also by tendons, joints, and ligaments. These structures and the muscles can also be affected by several factors early in life and cause lameness and latter impact bone development and strength. Sometimes, it is very difficult to differentiate which one was the primary cause, either the bone disorder or the gait pattern modification.
Bone development, the aetiology of disorders, and the influence of nutrition and management on the prevention of leg problems have been reviewed previously (Bradshaw et al., 2002; Oviedo-Rondón et al., 2006 a, b). This presentation will focus mainly on walking ability, which is more related to behavior, body conformation, the flexibility of the bird’s joints, and the strength of its muscles and tendons, than it is to bone disorders. Due to the complexity of the interactions among all of these factors, improving walking ability while maintaining desired body weight gain, remains a formidable challenge for poultry production. All husbandry, nutritional and even genetic selection methods that have been used to improve walking ability cause some reduction in growth rates; and, if that body weight gain reduction occurs at an early age, compensatory changes have not usually been seen at market ages. We must, therefore, develop a better understanding of the biomechanics of gait, behavior, and musculoskeletal development if we are to design better strategies for improving walking ability in our meat-type poultry.
Effects of walking ability
Locomotion involves smooth advancement of the body through space with the least mechanical and physiological energy expenditure. While the goal of walking is progression in the forward direction, limb motion is based on the need to maintain a symmetrical, low amplitude displacement of the center of gravity of the head and trunk in the vertical and lateral directions. Normal gait conserves both kinetic and potential energy and follows the principle of biological conservation of energy. Any lameness indicates a change in the “normal” gait pattern and increments in energy expenditure or reductions in activity (Muir et al., 1998; Waters and Mulroy, 1999).
Interruption of the normal gait cycle and the energy conserving characteristics of trunk and limb motion results in increased energy expenditure. Nevertheless, in response to a gait disability, heavy loads or unbalanced posture, an animal will adapt by performing compensatory gait substitutions to minimize the additional energy expenditure. Consequently, the gait patterns of heavy animals, such as can be observed in meat-type poultry, have to be different than seen in the lighter egg-type strains. The effectiveness and the associated penalties of those compensations depend upon the severity of the disability and the animal’s cardiovascular and musculoskeletal fitness. However, gait adaptations to compensate for heavy weight loads or unsound limbs are not always successful and can lead to problems in joints and finally lameness (Corr et al., 2003a, b, c; Buchwalder et al., 2004).
Although walking always requires energy expenditure, the ability to walk is absolutely necessary for the ability of meat-type birds to gain access to feed and water, to avoid pecking from other aggressive birds in the flock, and for the birds to have normal bone development, mineral density, bone strength, and to develop the frame they will need to support their muscle development. Bone architecture and biomechanical properties of bones change in response to the mechanical load and muscle strain placed upon them. On the other hand, joint loads result from several factors including bony alignment and gait biomechanics.
Adaptations of gait in unsound animals cause rotations of feet and every articulation that have consequences for the skeletal system, creating a progressive and degenerative problem. Reduced femoral torsion generally results in greater hip external rotation and an out-toed gait while those with more ante-torsion usually have greater hip internal rotation and a more in-toed gait. Limitations in hip mobility are associated with knee and hip osteoarthritis; however, it is unknown if it is primarily or secondarily related to the progression of the disease.
Osteoarthritis and other joint degenerative diseases that lead to pain are very common in commercial turkeys (Hocking et al., 2002b; Buchwalder and Huber-Eicher, 2005). Mild bone disorders by themselves do not have big impact on walking ability. The normal range for tibiotarsal torsion in birds is -5° to 20°, and mild torsions have no significant effect on gait (Corr et al., 2003a, b). Tibial dyschondroplasia incidence does not correlate with lameness or gait abnormalities in turkeys (Hocking et al., 2002a, b; Simsa et al, 2007 a,b) as it is observed in broilers. However, TD lesions can be a primary location for the development of osteomyelitis, which is the most common cause of longbone necrosis in turkeys. Additionally, the varus/valgus knee angle and the frontal plane moments that result during gait have been related to bone mineral distribution in the proximal tibia in healthy and diseased knees, implicating them in the pathogenesis of osteoarthritis. Internal tibial torsion (tibias with lower than normal external torsion angles) and low femoral ante-torsion angles have been linked with the incidence and severity of knee osteoarthritis. Arthritis and tendon weaknesses can also be initiated by early bacterial, viral and mycoplasma infections.
Energy cost of walking
It is important to remember that the normal walking activity in birds has an influence on dietary energy requirement variability between flocks and among birds in a flock. Energy requirements are often estimated under restricted room conditions, often in respiration chambers or metabolic cages, which underestimate the requirements for birds raised on the ground, so for practical applications these values must be corrected. For example, Johnson and Farrell (1983) and Spratt et al. (1990) (cited by Sakomura, 2004) estimated energy requirements of 87.24 and 87.71 kcal/kg0.75/day, respectively, for broiler breeder hens in metabolic chambers at 21°C. A similar result (91 kcal/kg0.75/day) was observed for breeders in cages at 21°C, while for those raised on the ground the energy requirement was 113 kcal/kg0.75/day (Sakomura, 2004).
The energy requirement for activities is about 50% of basal metabolism and is influenced by raising conditions. Birds in cages present lower activity and heat production, about 30% of basal metabolism. Sakomura (2004) reports that total energy requirement for maintenance of broiler breeders raised on the ground is 20% higher than for those reared in cages. In her studies, broiler breeder hens raised on the ground showed higher heat production (144.18, 135.18, 136.56 kcal/kg0.75/day) than those raised in cages (77.83, 65.20, 59.19 cal/kg0.75/day) at 13, 21 and 30°C, respectively. The expenditure of energy for activities of laying hens is about 20 to 25% of heat production (MacLeod et al., 1982).
The energy unit in metabolic studies is the gram/calorie (cal) or the kilogram-calorie (Kcal). Direct measurement of heat production/calorimetry in exercising subjects is impractical. Caloric consumption is therefore calculated indirectly from data on the volume of O2 consumption and CO2 production. It is considered that 5 calories equals 1 ml O2.
The terms power and work are utilized to describe energy expenditure. The power requirement (rate of O2 consumption) is the milliliters of O2 consumed per kilogram body weight per minute (ml:kg per min). Physiological work is the amount of energy required to perform a task. Physiological work (O2 cost) during level walking is the amount of oxygen consumed per kilogram body weight per unit distance traveled (ml: kg.per m). The O2 cost is determined by dividing the power requirement (rate of O2 consumption) by the speed of walking. By comparing the energy cost of pathological gait to the corresponding value for normal walking, it is possible to determine the gait efficiency. The rate of O2 consumption relates to the level of physical effort and the O2 cost determines the total energy required to perform the task of walking. O2 cost may be elevated by either an increased O2 rate or by a low walking speed with a normal rate of O2 consumption. Depending on the degree of lameness, the additional energy expenditure for locomotion can be increased by 20% and up to 60% (Waters and Mulroy, 1999).
Walking in Broilers and Turkeys
Chickens and turkeys exert two-peaked vertical forces on the ground when walking, and single-peaked forces when running. Very young chicks (i.e., 0–3 days old) naturally walk with shorter stride lengths, spend more time in contact with the ground, and more time supported by two legs compared with older chicks (10–14 days old) (Muir et al., 1996). Walking ability is lower in fast-growing birds, independently of diet or management. This different behavior actually starts very early in life. Bizeray et al. (2000) observed that at 2 and 3 days, the activity of broilers was half that of label rouge chicks during standing bouts. The duration of walking per bout was 19±4 s for broilers and 45±4 s for label rouge (P<0.05). The correlations between the levels of activity at early and later ages suggest that selection of young mobile broiler chicks might increase activity at a later age and might therefore reduce the occurrence of leg abnormalities or improve average walking ability in flocks.
Manipulation of locomotor experience in chicks can alter their locomotor pattern (Muir and Chu 2002). When exercise restriction is imposed on chicks immediately after hatching for a minimum of 6 days, they move with shorter stride lengths and make smaller head excursions during head bobbing compared with control animals (Muir and Chu 2002). Head bobbing, which occurs during walking in birds, is an optokinetic response, driven by the movement of the visual world across the retina (i.e., optic flow). During walking, the movement of the visual world is generated by forward translation, and thus the relationships between locomotor parameters and the extent of head excursions could provide important insights into the control and development of head bobbing. For example, the shorter stride lengths that occurred in exercise-restricted animals might have accounted for the smaller head excursions seen in the same group of animals because stride length determines the horizontal distance moved and thus the amount of retinal slip occurring for each stride. Stride length and head excursions are, in fact, moderately correlated (Muir and Chu, 2002). Nevertheless, other factors in addition to stride length must influence the extent of head excursions during posthatching development because head excursions continued to increase for >10 days after hatching, whereas stride length ceased to change after 4 days (Muir and Gowri, 2005). Consequently, the availability of space and general management to guarantee comfort during the early brooding phase may impact the walking ability of birds.
Resch-Magras et al. (1993) carried out a comparative locomotion analysis of healthy and lame male commercial turkeys. Walking turkeys were filmed from both right and left lateral views. Video films were analyzed. The coordination and the succession of hind limb movements were studied with time and space components. The gait of healthy turkeys was a walk with perfectly symmetrical and repeatable hind limb movements. All lame turkeys' movements showed abnormalities in space-time coordination. These abnormalities were bilateral but non-symmetrical, intermittent, and non-systematic. Their intensity increased with increase in the degree of clinical handicap. However, in all lame turkeys, some degree of an abnormal tibial movement was observed, i.e. they had a shorter amplitude of forward movement with variable intensity.
Predisposing Factors that Affect Walking Ability in Poultry
Several factors cause changes in normal walking patterns in turkeys and broilers:
- Changes in body conformation caused for genetic selection for fast-growth and broad breast
- Hypothyroidism observed in birds selected for fast growth and lower physical activity
- Increased developmental fluctuating asymmetry when facing environmental stress
- Modifications in tendon development due to lack of exercise, aging or nutrition
- Pain due to secondary osteoarthritis and other degenerative joint diseases
- Spinal cord injuries and vertebra deviations (kyphosis or scoliosis)
- Management factors such as lighting, stocking density, distance between feeders and drinkers, and environmental conditions.
- Infections of joints by bacteria (Streptococcus, E. coli, Salmonella), mycoplasmas, and tendon infections with reovirus, that although are not reported in turkeys, may happen.
Body conformation
Genetic selection for a heavier broad-breasted turkey has increased the instability during walking that is compensated for by changes in gait cycle. Abourachid (1991) demonstrated the incremental stresses on the pelvic muscles of broad-breasted turkeys selected for several generations in comparison with non selected birds. The stresses are induced by the anterior position of the centers of gravity of their trunks as result of selected pectoral hypertrophy.
Abourachid (1993) did not observe significant differences between the gaits of traditional and broad-breasted strains from the lateral views. However, the posterior views of the walk of both strains show lateral oscillations, which are very slight in the traditional strain but very marked in the broad-breasted turkey. In the latter, modification of the centre of gravity may increase the energetic cost of walking and be associated with problems of lameness. Similar data has been obtained by Corr et al. (2003a) in chickens and hens. Havenstein et al. (2004a, b) reported that turkey breast muscle increased by approximately 6.5%, whereas thigh muscle yield increased by only 0.65% between 1966 and 2003, due to commercial breeder selection practices. The relative proportion of total body live weight in the femur bone has actually decreased by 0.35% during that time period. The associated disproportional increase of the breast muscle (25 – 30% of the body weight) compared to other muscles, tibia and femur bone leads to a physiologically unbalanced weight distribution (Figure 1).
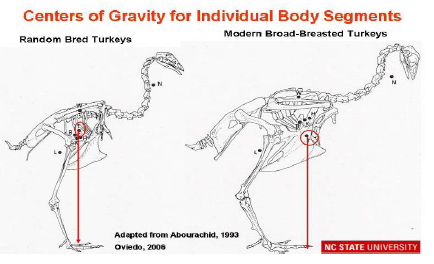
Femur and tibia bones and joints are stressed excessively in broad-breasted turkeys and broilers when compared with random bred strains, as is shown in Figure 2 (Abourachid, 1993). The femur’s abduction reduces the length of its projection on the sagittal plane and keeps the knee’s projection in this plane more posterior. The moment arms of the weight on the pelvic joints are subsequently larger. As the locomotor apparatus is geometrically similar, the required effort to maintain balance is increased; the stress over the extensor muscles in broad-breasted turkeys and fast-growing broilers is higher than in non-selected strains (Abourachid, 1993). This leads to modifications of motion sequences (Corr et al., 2003b).To compensate for these changes in body structure, turkeys walk slowly with their toes pointed outwards, taking short, wide steps (Corr et al., 2003b); and, with aging they become less active (Kestin et al., 2001). Broad-breasted fattening turkeys reduce the time they are active i.e. standing and walking and consequently putting weight on their legs.
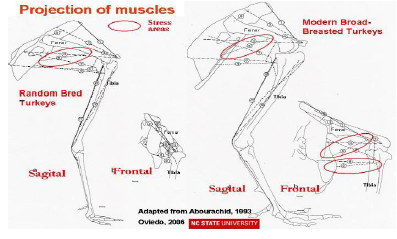
Three selection approaches can be taken to genetically improve walking ability in turkeys and broilers, either to improve leg bone mass and strength, to increase leg muscle development or change posture (Nestor and Emmert, 1990; Abourachid, 2000). Since, shank width is highly heritable and is strongly correlated with both shank weight and the weight of the tibiotarsal and femur bones, selection for shank width could provide a mean to improve walking ability. However, when a turkey line selected for several generations for shank width was used to improve commercial lines, the slight improvement noted in walking ability of the male crosses was offset by the reduction in body weight and breast width of the crosses relative to the pure commercial sire lines (Nestor and Anderson, 1998). Thus, this alternative was proven to not to be feasible for improving the walking ability. Direct selection for leg muscle mass, or specific muscles, or even functional adaptation in upright postures may provide another alternatives.
Hypothyroidism
Selection for fast growth and improved feed utilization has also resulted in commercial poultry strains have lower thyroid activity compared to non-selected birds (Gonzales et al., 1999). Management factors and heat stress can increase this hypothyroidism in some birds within commercial flocks (Oviedo-Rondón et al., 2006a). Hypothyroid animals exhibit lower physical activity, weakness of limbs and leg trembling when walking (Zoeller and Rovet, 2004). This metabolic factor may affect broilers and turkeys as well, and its relationship with walking ability should be evaluated. In fact, broilers spend 75 to 85% of their time lying down, and it is very difficult to make them increase their physical activity (Bizeray et al., 2002).
Fluctuating asymmetry, physical activity and bone development
Genetic selection for fast growing birds and environmental stresses increase the asymmetry between the right and left limbs in length, size and development (Møller and Manning, 2003). At NC State University we have observed that elevated temperatures and hypoxia in the hatcher cause this asymmetry in hatching poults (Oviedo-Rondón et al., 2007). During our research at NC State University, we have also evaluated the normal gait patterns of turkeys including animals with the lameness problem known as valgus, varus, crooked toes, and shaky legs, using a pressure sensitive walkway (Oviedo-Rondón et al, 2006). We measured peak vertical force, vertical impulse, peak contact area, foot contact time and stride length. This pilot project demonstrated for the first time that some leg conditions can create asymmetry in walking forces that may impact asymmetrical bone development in each leg. Asymmetrical bone development is compensated for by asymmetric gait which can cause future lameness (Resch-Magras et al., 1993). Currently, we are evaluating the development of normal and abnormal gait, differences among turkey strains, and its influence on bone biomechanical properties around market age (Figure 3).
Tendon development
Roberts and Scales (2002, 2004) studied the mechanical power output, muscle function, joint work and energy cost of bipedal running in wild turkeys and observed the importance of muscle and tendon structure and function that produce force while minimizing mechanical work. During walking and running, the tendons and aponeurosis of legs absorb lengthening of the whole muscle-tendon unit, limiting further tendon deformation, thereby allowing the muscle fibers to function isometrically, or to actually shorten. This mechanism has been reported to preserve significant quantities of muscular energy in the locomotion process. Elastic recoil is principally associated with the tendons, and it converts most of the stored energy into kinetic energy as the foot of the animal leaves the ground. It also reduces tendon strain from a given stress, and it provides greater load-bearing capacity to the tissue.
In the lateral gastrocnemius muscle of the turkey, direct measurements of force and fiber length have shown that the active muscle produces high force but little work while the tendon produces much of the work because of elastic deformation and recovery. Unlike mammalian flexor tendons, which undergoes increased cross-linking with age, segments of the turkey gastrocnemius tendon apparently bear increasing loads by mineralizing during aging (Landis and Silver, 2002). Mineralization has been suggested as an effective and important means for preserving elastic energy storage while providing the increased ability for load bearing needed in adult bird locomotion. In this manner, energy produced during turkey gastrocnemius muscle contraction can be efficiently, instantaneously stored in the proximal mineralizing segments of this tendon and can then be transmitted through the distal non-mineralized segment as required for locomotory function and response.
Mineralization of the tendons in the legs and wings of certain birds are normal. Tendons are not initially mineralized, but undergo progressive calcification from 10–12 weeks of age in turkeys, along the distal to proximal direction, probably as a result of energy storage requirements and a physiological stress-induced calcification. The distal rounded, thick gastrocnemius bifurcates into two smaller proximal segments that mineralize with time. Mineral deposition occurs at or near the bifurcation, proceeding in a distal-to-proximal direction along the segments toward caudal and medial muscle insertions of the bird’s hip. This mineral formation appears mediated first by extracellular matrix vesicles and later by type I collagen fibrils. Biomechanical analyses indicate lower tensile strength and moduli for the thick distal gastrocnemius compared to narrow, fan-shaped proximal segments. Tendon mineralization here appears to be straininduced, the muscle forces causing matrix deformation leading conceptually to calcium binding through the exposure of charged groups on collagen, release of sequestered calcium by proteoglycans, and increased diffusion.
In order for normal tendon development to occur, the bird has to exercise its legs (Foutz et al., 2007a, b). Foutz et al. (2007b) observed that without load bearing, the structural strength and toughness of the gastrocnemius tendon of broilers were reduced by 10 and 30%, respectively. Decreased activity in some birds may reduce tendon strength, change biochemical and biomechanical properties and cause additional difficulties in the walking ability in these birds as they age (Foutz et al. 2007a, b; Moussa et al. 2007). Studies of tendon development and its biomechanical properties among commercial lines of birds are scarce, and are needed if we are to understand the causes for the progressive reduction in walking ability of meat-type poultry.
Pain
Degenerative joint diseases are common in turkeys and can cause severe and chronic pain, especially when weight is put on legs (Buchwalder et al., 2004). Corr et al. (2003) observed that the synovial fluid of fast-growing broilers fed ad-libitum was more turbid and had thicker synovial membranes than restricted-fed broilers. The synovial fluid from lame broilers contained more heterophils, indicating an inflammatory arthropathy.
In birds, the reactions to pain are subtle and are limited to slight alteration in posture, changes of perching position, decreased appetite and/or a general reduction in activity (Paul-Murphy and Ludders, 2001). The most common assessment method to evaluate pain is to measure behavior in the presence or absence of analgesia. Buchwalder et al. (2004) evaluated the effects of the analgesic butorphanol on the activity behaviors in turkeys. At week seven, the treated birds spent significantly more time putting weight on their legs than did the control birds. At week 12, the same tendency was observed. No significant differences were found in the distances covered by the animals. The authors concluded that fast growing turkeys reduce the time they are putting weight on their legs because these behaviors may be associated with pain in their joints. Buchwalder et al. (2004) also mentioned that foot pad dermatitis as a source of pain can be excluded. These same conclusions have been obtained in studies with broilers (Geown et al., 1999; Danbury et al., 2000; Hocking et al., 2001 cited by Buchwalder et al. 2004) and turkeys (Duncan et al., 1991; Hocking et al., 1999, cited by Buchwalder et al., 2004) using other analgesics.
3.6 Spinal cord injuries and vertebra deviations
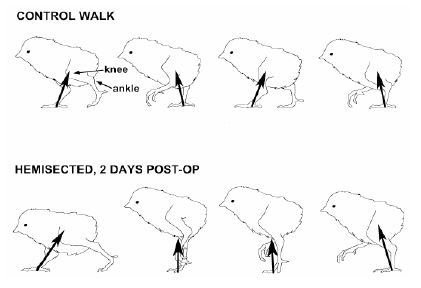
Avian thoracic vertebrae are fused except at the joints proximal and distal of T-6. This free-moving vertebra can become ventrally deviated (kyphosis) or laterally deviated (scholiosis) or both. Turkeys and broilers with these lesions have a “humpback” posture and also may have “shaky leg” behavior. The problem is more commonly observed after 12 weeks of age. The deformation of the vertebral channel can impinge the spinal cord and may affect gait. Muir et al. (1998) demonstrated that chicks can have adaptive gait responses to incomplete injury of the spinal cord (Figure 4). This can demonstrate that hatchlings that suffer injuries during manipulation in the hatchery or during transportation may survive and just adapt their locomotor patterns.
Nutrition and Management to Improve Walking Ability
A good way to improve walking ability is by enhancing locomotor activity of the flock. The husbandry methods that have had success to enhance activity in flocks include decreasing stocking density (Febrer et al., 2006), increasing distance between feeders and drinkers, or modifying feed availability by changes in schedules of distribution, feeding alternating different diets during the day (sequential feeding) that include variation on nutrient concentration or ingredient composition or form (whole wheat and complete diets) (Bizeray et al., 2002). Reducing protein or amino acid levels, nutrient density or energy levels, or changing feed form from pellets to mash can reduce bone disorders, cause small improvements in gait scores, but rarely enhance walking activity (Yalçin et al., 1998; Hocking et al., 2002a; Brickett et al., 2007). The benefits of these methods are always correlated with a reduction in growth rate that in most occasions does not have compensatory gains.
Bone development and bone strength can be improved by nutritional methods without reducing body weight gain or feed efficiency, but to improve walking ability of some individuals, it is almost always necessary to reduce the average body weight gain of the population.
Lighting programs have had variable results to affect gait scores, reduce leg problems in general or enhance activity in flocks (Sørensen et al., 1999; Kristensen et al., 2006; Olanrewaju et al., 2006; Bricket et al., 2007). The successful lighting programs generally cause a reduction in growth rate.
Conclusions
It is important that we improve the walking ability of commercial poultry flocks because it will improve the efficiency of energy utilization and will help to avoid bone disorders. Walking ability of the majority of birds in a flock is mainly related to body conformation, flexible joints, strong tendons, behavior and cognition. Severe bone disorders may affect walking ability of specific birds, but most frequently changes in “normal” gait patterns may affect bone development. Combinations of management and nutritional methods to stimulate walking activity and reduce growth rates can be used to maintain adequate walking ability in poultry flocks. However, it is important to keep in mind that the success of these methods is limited. Future genetic selection methods should be based on biomechanical parameters of gait, and knowledge of tendon and muscle development.
References:
Abourachid, A. 1991. Comparative gait analysis of two strains of turkey, Meleagris gallopavo. Br. Poult. Sci. 32(2):271-277.Abourachid, A. 1993. Mechanics of standing in birds: functional explanation of lameness problems in giant turkeys. Br. Poult. Sci. 4(5):887-98.
Abourachid, A. 2000. Bipedal locomotion in birds: the importance of functional parameters in terrestrial adaptation in Anatidae. Can. J. Zool. 78:1994-1998.
Bizeray D., C. Leterrier, P. Constantin, M. Picard and J.M. Faure. 2000. Early locomotor behaviour in genetic stocks of chickens with different growth rates. Applied Animal Behaviour Science. 68:231-242.
Bizeray D., C. Leterrier, P. Constantin, M. Picard and J.M. Faure, 2002. Sequential feeding can increase activity and improve gait score in meat-type chickens. Poult. Sci. 81:1798-1806.
Bradshaw, R.H., R. D. Kirkden and D.M. Broom. 2002. A review of the aetiology and pathology of leg weakness in broilers in relation to welfare. Avian Poult. Biol. Rev. 13, 45-103.
Brickett, K.E., J.P. Dahiya, H.L. Classen, C.B. Annett and S. Gomis. 2007. The impact of nutrient density, feed form, and photoperiod on the walking ability and skeletal quality of broiler chickens. Poult. Sci. 86:2117-2125.
Buchwalder T. and B. Huber-Eicher. 2005. Effect of the analgesic butorphanol on activity behavior in turkeys (Meleagridis gallopavo). Res. Vet. Sci. 79:239-244.
Buchwalder, T. 2004. Effect of familiarity, group size, and floor space availability on agonistic behaviour in fattening turkeys (Meleagris gallopavo) and effect of an analgesic on their activity behaviour. Doctoral Dissertation Universität Bern.
Corr, S.A., M.J. Gentle, C.C. McCorquodale and D. Bennett. 2003a The effect of morphology on the musculoskeletal system of the modern broiler. Anim. Welf. 12, 142-157.
Corr, S.A., M.J. Gentle, C.C. McCorquodale and D. Bennett. 2003b. The effect of morphology on walking ability in the modern broiler: a gait analysis study. Anim. Welf. 12, 159-171.
Corr, S.A., M. Maxwell, M.J. Gentle and D. Bennett. 2003c. Preliminary study in joint disease in poultry by the analysis of synovial fluid. Veterinary Record 152: 549-554.
Febrer, K., T.A. Jones, C.A. Donnelly and M.S. Dawkins. 2005. Forced to crowd or choosing to cluster? Spatial distribution indicates social attraction in broiler chickens. Animal Behaviour 72:1291-1300.
Foutz T.L., A.K. Griffin, J.T. Halper and G.N. Rowland. 2007a. Effects of activity on avian gastrocnemius tendon. Poult. Sci. 86(2): 211-218.
Foutz T., A. Ratterman and J. Halper. 2007b. Effects of immobilization on the biomechanical properties of the broiler tibia and gastrocnemius tendon. Poult. Sci. 86(5): 931-936.
Gonzales, E., J. Buyse, J.R. Sartori, M.M. Loddi and E. Decuypere. 1999. Metabolic disturbances in male broilers of different strains. 2. Relationship between the thyroid and somatotropic axes with growth rate and mortality. Poult. Sci. 78, 516-521.
Hocking, P.M., G.W. Robertson and C. Nixey. 2002a. Effects of dietary calcium and phosphorus on mineral retention, growth, feed efficiency and walking ability in growing turkeys. Br. Poult. Sci. 43:607-614.
Hocking, P.M., S. Wilson, L. Dick, L.N. Dunn, G.W. Robertson and C. Nixey. 2002b. Role of dietary calcium and available phosphorus in the aetiology of tibial dyschondroplasia in growing turkeys. Br. Poult. Sci. 43:432-441.
Kristensen H.H., G.C. Perry, N.B. Prescott, J. Ladewig, A.K. Ersboll and C.M. Wathes. 2006. Leg health and performance of broiler chickens reared in different light environments. Br. Poult. Sci. 47(3):257-263.
Landis, W.J. and F.H. Silver. 2002. The structure and function of normally mineralizing avian tendons. Comparative Biochemistry and Physiology - Part A: Molecular & Integrative Physiology. 133 (4):1135-1157
McLeod M.G., T.R. Jewitt, J. White, M. Verbrugge and M.A. Mitchell. 1982. The contribution of locomotor activity to energy expenditure in the domestic fowl. In Energy metabolism of farm animals (Ekern A. and F. Sundstøl, eds.) European Association for Animal Production EAAP Publication No. Page 297-300.
Mench, J. 2004. Lameness. In: Weeks, C. and Butterworth, A. (eds.), Measuring and Auditing Broiler Welfare, p3-17. CAB International.
Møller, P.A. and J. Manning. 2003. Growth and developmental instability. The Veterinary Journal 166:19-27.
Moussa, M., R. Babilé, X. Fernandez and H. Rémignon. 2007. Biochemical and biomechanical properties of tendons in two commercial types of chickens. Animal 1(7):983-988.
Muir G.D., J.M. Gosline and J.D. Steeves. 1996. Ontogeny of bipedal locomotion: walking and running in the chick. J. Physiol. 493:589-601.
Muir G.D., S.L. Katz, J.M. Gosline and J.D Steeves. 1998. Asymmetric bipedal locomotion – an adaptive response to incomplete spinal injury in the chick. Exp. Brain Res. 122:275-282.
Muir G. D. and T. K. Chu. 2002. Posthatching locomotor experience alters locomotor development in chicks. J Neurophysiol. 88: 117-123.
Muir, G.D. and K.S.V. Gowri. 2005. Role of motor and visual experience during development of bipedal locomotion in chicks. J Neurophysiol. 94: 3691-3697.
Nestor, K.A. and J.W. Anderson. 1998. Effect of crossing a line selected for increased shank width with two commercial sire lines on performance and walking ability of turkeys. Poult. Sci. 77 (11):1601-1607
Nestor, K.E. and D.A. Emmerson. 1990. Genetics of leg strength in turkeys. National Breeders' Roundtable, St. Louis, Missouri.
Olanrewaju, H.A., J.P. Thaxton, W.A. Dozier III, J. Purswell, W.B. Roush and S.L. Branton. 2006. A Review of lighting programs for broiler production. International Journal of Poultry Science 5 (4): 301-308.
Oviedo-Rondón, E.O., J. Small, M.J. Wineland, V. Christensen, D. Ort, M. Mann and S.V.L. Funderburk, 2007. Temperature and oxygen conditions during the plateau stage of incubation affect long bone development in broilers and turkeys. IPSF-Abstracts.
Oviedo-Rondón, E.O., P.R. Ferket and G.B. Havenstein. 2006a. Nutritional factors that affect leg problems in broilers and turkeys. Avian and Poultry Biology Reviews 17(3):89-103.
Oviedo-Rondón, E.O., P.R. Ferket and G.B. Havenstein. 2006b. Understanding long bone development in broilers and turkeys. Avian and Poultry Biology Reviews 17(3):77-88.
Oviedo-Rondón, E. O., P.R. Ferket, P.L. Mente, B.D.X. Lascelles, H.J. Barnes, D.V. Bohórquez and J.L. Grimes. 2006c. Biomechanical Factors that Influence Spiral Femoral Fractures of Turkeys. AVMA-AAAP.
Paul-Murphy, J. and J. Ludders. 2001. Avian analgesia. Veterinary Clinics of North America: Exotic Animal Practice 4(1) 35-45.
Resch-Magras, C., Y. Chérel, M. Wyers and A. Abourachid. 1993. Analytical study of the locomotion of the meat turkey cock. Comparison between sound turkeys and turkeys with locomotor problems. Vet Res. 1993;24(1):5-20.
Roberts, T.J. and J.A. Scales. 2002. Mechanical power output during running accelerations in wild turkeys. Journal of Experimental Biology 205: 1485-1494
Sakomura N.K. 2004. Modeling energy utilization in broiler breeders, laying hens and broilers Rev. Bras. Cienc. Avic. 6(1). Campinas, Jan./Mar. 2004.
Shalev, B.A. and H. Pasternak. 1998. The relative energy requirement of male vs female broilers and turkeys. Poult. Sci. 77:859-863.
Simsa, S., A. Hasdai, H. Dan and E. Monsonego-Ornan. 2007a. Differential regulation of MMPs and matrix assembly in chicken and turkey growth-plate chondrocytes. Am. J. Physiol. Regul. Integr. Comp. Physiol. 292: R2216-R2224.
Simsa, S. A. Hasdai, H. Dan and E.M. Ornan. 2007b. Induction of tibial dyschondroplasia in turkeys by tetramethylthiuram disulfide (Thiram). Poult. Sci. 86(8): 1766-1771.
Sørensen, P, G. Su and S.C. Kestin. 1999. The effect of photoperiod:scotoperiod on leg weakness in broiler chickens. Poult. Sci. 78:336-342.
Waters, R.L. and S. Mulroy. 1999. The energy expenditure of normal and pathologic gait. Gait and Posture 9:207-231.
Yalçin, S., P. Settar and O. Dicle. 1998. Influence of dietary protein and sex on walking ability and bone parameters of broilers. Br. Poult. Sci. 39:251-256.
Zoeller, R.T. and J. Rovet. 2004. Timing of thyroid hormone action in the developing brain: clinical observations and experimental findings. Journal of Neuroendocrinology 16:809-818.
February 2009








