



Responsiveness of High Screenings and Commercial Cereal Grains to a Blend of Xylanase and Phytase Enzyme Products
The enzyme blend improved apparent metabolisable energy values for some wheats and only one barley but it had little or no effect on other types of grain tested, according to a paper presented at the 2011 Australian Poultry Science Symposium by R.J. Hughes of SARDI, M.S. Geier, J.L. Black, A.M. Tredrea, S. Diffey and S.G. Nielsen.
Summary
Two experiments evaluated the apparent metabolisable energy (AME) values for cereal grains fed with and without a blend of xylanase and phytase enzyme products to broiler chickens.
The first experiment involved 30 high-screenings grains (10 wheats, 11 barleys, 3 triticales, and 6 sorghums). The second experiment involved 50 grains (20 wheats, 13 barleys, three triticales and 14 sorghums) donated by commercial companies throughout Australia.
Each experiment included ‘connectivity’ grains used in previous experiments that contributed to the AusScan near infrared reflectance (NIR) calibration database. The inclusion of ‘connectivity’ grains (15 in Experiment 1 and 17 in Experiment 2) enabled data to be analysed statistically for valid comparison across many experiments conducted in the period 1998 to 2010. These grains were also fed with and without the blend of enzymes.
In general, the results show that the enzyme blend improved AME values for some wheats and only one barley, and had little or no effect on other types of grain.
Introduction
Black et al. (2009) described calibrations based on NIR spectroscopy for estimating the AME content and AME Intake Index of cereal grains for broiler chickens. The calibrations were developed from results obtained in the Premium Grains for Livestock Program. Updated NIR calibrations were reported by Black et al. (2010). These new calibrations included results from an extra 55 grains, comprising 30 high screenings grains and 25 grains donated by industry. This greatly improved the ability to predict values for unknown grains and the accuracy of prediction. Further improvements are anticipated when results from an additional 25 industry grains are included in the database.
Two large AME experiments funded by RIRDC Chicken Meat were required to evaluate a total of 80 grains, which included 30 high screenings grains (Experiment 1) and 50 industry grains (Experiment 2). All grains were fed with and without a blend of xylanase and phytase enzyme products. This paper describes the responses in grain AME to enzyme products.
Materials and Methods
The AME values of grains were determined across a series of conventional energy balance studies involving measurements of feed intake and excreta output as described by Mollah et al. (1983) with minor modifications, and subsequent measurement of gross energy values of feed and excreta by bomb calorimetry.
High screenings and other weather-damaged samples (total 30 comprising 10 wheats, 11 barleys, three triticales and six sorghums) collected as part of a Pork CRC project were examined in Experiment 1. The second experiment involved 50 grains (20 wheats, 13 barleys, three triticales and 14 sorghums) donated by commercial companies throughout Australia.
Experiment 1 required four batches of day-old feather-sexed broiler chickens, while Experiment 2 used eight batches. Each batch was raised in floor pens on a commercial broiler diet to 22 days of age and then transferred in single-sex groups of five to metabolism cages in controlled temperature rooms. Air temperature was maintained at 26°C at the start of the seven-day experiment and lowered daily until it was 23°C at the end. Experimental diets contained grain, casein, dicalcium phosphate, limestone, DL-methionine, mineral and vitamin premix, salt and choline chloride. All grains were fed with and without a blend of xylanase (Porzyme 93010 at 50g per tonne for wheat, barley and triticale, or Rovabio Excel at 200g per tonne for sorghum) and phytase (Phyzyme TPT at 50g per tonne) enzyme products.
Dietary treatments were replicated four times (two cages of males and two cages of females). Cold-pressed diets were fed for seven days. The first three days enabled the chickens to adapt to the feeds. During the following four days, all excreta were collected and dried at 85°C. Feed intake was measured during the adaptation and collection phases of the study. Birds were weighed at the start and end of the seven-day period. Dry matter contents of samples of pelleted and milled feeds were measured. Gross energy values of dried excreta and milled feeds were measured with a Parr isoperibol bomb calorimeter. AME of the grain was calculated by subtracting from the total energy intake the energy contribution of casein, which was assumed to be 20.1MJ per kg dry matter (Annison et al., 1994).
Results
In Experiment 1, there were significant two-way interactions between grain type and enzyme (P<0.001) and grain type and sex (P<0.05) on AME values for high screenings grains (Table 1). The effects of enzyme and sex on individual wheats are summarised in Figure 1. The proportion of grains passing through a 2-mm sieve averaged 17 per cent and ranged from 1.4 per cent for wheat 1753 and 53.1 per cent for wheat 1762 in Figure 1. Regression analysis indicated AME values for grain declined by about 0.2MJ per kg for each 10 per cent rise in screenings, in both male and female chickens, when given enzymes. In the absence of enzyme, the responses to screenings were highly variable, particularly for male chickens.
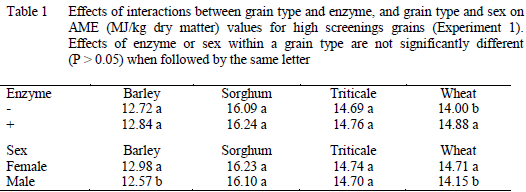
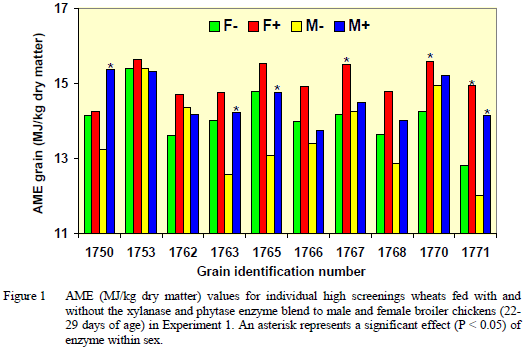
In Experiment 2, there was a significant three-way interaction between individual grains, enzyme and sex. Enzymes improved AME for only one sample of barley fed to male chickens. The effects of enzyme and sex on individual wheats are summarised in Figure 2.
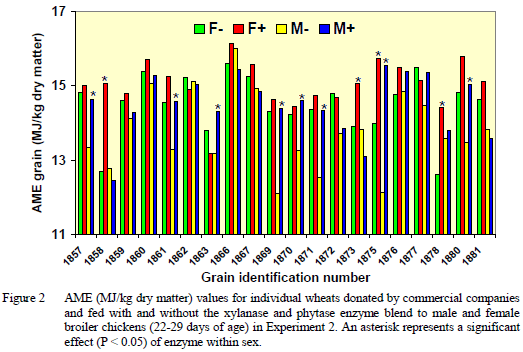
The soluble non-starch polysaccharide (NSP) values (in g/kg dry matter) for wheat ranged from 9.8 for wheat 1876 to 15.2 for wheat 1860 in Figure 2, and averaged 12.1. These soluble NSP values are considerably lower than 20 g/kg dry matter normally found in “low AME” wheats (Choct et al., 1996). The insoluble non-starch polysaccharide values (in g/kg dry matter) for wheat ranged from 70 for wheat 1861 to 115 for wheat 1878 in Figure 2, and averaged 83. Regression analysis indicated negligible change in AME values for grains due to soluble NSP level, however, for each 10g per kg increase in insoluble NSP, AME declined by about 0.3 and 0.4MJ per kg dry matter, respectively, for males and females given enzymes.
Discussion
The results of both experiments showed that the blend of xylanase and phytase enzymes improved AME values for some but not all wheats, and only one sample of barley. Other types of grain were unaffected. Variable responses of different wheats to enzymes were observed in high screenings grains with presumably higher than usual concentrations of soluble and insoluble NSP. Increasing concentrations of insoluble NSP, but not soluble NSP, depressed AME values for wheat sourced from commercial feed mills. The lack of response to this enzyme blend in some wheats (high screenings and industry-sourced) probably reflects differences in cell wall matrices in grains and the way NSP are bound to other cell wall components (Choct et al., 1996), with net effects of xylanase and phytase in the enzyme blend differing between grains. These experiments were not designed to distinguish between the effects of xylanase and phytase components of the blend, but the respective effects are likely to be additive in wheat diets (Cowieson and Bedford, 2009).
The results of both experiments show that males fed barley and wheat can have lower AME values than females, whereas there were no differences between males and females given triticale and sorghum. These results are consistent with earlier work by Hughes et al. (2001) who found similar reductions in AME of wheat and barley fed to male chickens, but no differences when fed triticale or sorghum. This phenomenon may indicate that the β-glucan and arabinoxylan NSP affect males more so than females. Hughes (2003) conjectured that sex differences may have arisen through establishment of different gut microflora profiles as a result of differential flow of undigested nutrients into the hindgut acting as differential growth media for bacteria, or by exchange of chemical messages between host tissue and gut microflora. These possibilities warrant further investigation.
In conclusion, the results show that the particular blend of xylanase and phytase enzymes improved AME values for some, but not all wheats, and only one sample of barley. Other types of grain were unaffected by enzymes. Significant responses to enzymes in wheat diets were not always associated with high NSP levels.
Acknowledgments
The RIRDC Chicken Meat Programme provided financial support (Project PRJ-002973). The Pork CRC provided high screenings grains. The authors thank Mrs Shuyu Song, University of New England, Armidale NSW for NSP analyses.
References
Annison G., Choct M. and Hughes R.J. (1994) Proceedings of Australian Poultry Science Symposium, 6:92-96.
Black J.L., Hughes R.J., Nielsen S.G., Tredrea A.M. and Flinn P.C. (2009) Proceedings of Australian Poultry Science Symposium, 20:31-34.
Black J.L., Hughes R.J., Geier, M.S., Nielsen S.G., Tredrea A.M. and Flinn P.C. (2010) Proceedings of Australian Poultry Science Symposium, 21:51-54.
Choct M., Hughes R.J., Trimble R.P., Angkanaporn K. and Annison, G. (1996) Journal of Nutrition, 125: 485-492.
Cowieson A.J. and Bedford M.R. (2009) World’s Poultry Science Journal, 65: 609-624.
Hughes R.J. (2003) Proceedings of Australian Poultry Science Symposium, 15: 172-176.
Hughes R.J., Choct M. and van Barneveld R.J. (2001) Proceedings of Australian Poultry Science Symposium, 13:30-38.
Mollah Y., Bryden W.L., Wallis I.R., Balnave D. and Annison E.F. (1983) British Poultry Science, 24: 81-89.
Further Reading
| - | You can view other papers presented at the Australian Poultry Science Symposium 2011 by clicking here. |
July 2011








