Technically Speaking
Combating Coccidiosis in Broiler Breeders
 Charles L. Hofacre, DVM,
MAM, PhD
The University of Georgia
|
How many times have you
heard someone in the poultry
industry say, "We've been
doing it this way for 25 years" ?
Even if some things have remained
the same, 25 years has meant a lot of
change in the broiler industry, especially
regarding genetic progress.
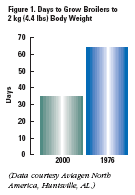
Consider broilers in 1976. It took about
65 days to reach 4.4 lbs (2 kg) body
weight. Today, broilers reach that
weight in only 35 days (Figure 1).
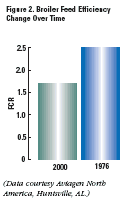
Broilers today are also more feed efficient:
1.70 now vs. 2.50 in 1976 (Figure
2).
Improvements in efficiency due to
genetic progress have a price, however.
It's more difficult to manage breeders
now because they grow so fast and
are so feed-efficient. If anything limits
their early growth, such as poor chick
quality, poor brooding conditions or
disease, the result is a flock with poor
body weight and uniformity of frame
size.
Impact on growth
Coccidiosis is one disease that can
greatly affect growth at its most critical
stage for frame size development.
In today's highly competitive broiler
industry, we strive to capitalize our pullets
by feeding the least amount necessary,
which places emphasis on frame
size. The majority of skeletal growth in
broiler breeders occurs in the first 5 to
6 weeks of life.1 If birds experience
either clinical or subclinical coccidiosis
during this period, their frame size -
and thus flock uniformity "will be
greatly affected (see photo).
In a flock with poor uniformity,
hens that are smaller become timid and
fall even farther behind. It is easy for us
to see the impact on egg production,
but we often don't realize that roosters
in the same house with pullets are also experiencing coccidiosis.
Coccidiosis can become an even
greater problem in male chicks since
they usually are smaller than females; if
they don't surpass female weight by
50% by 5 weeks of age, we risk having
poor hatchability for the life of the
flock.
The biology of coccidia
In general, the life cycle of all coccidia are similar. The bird eats a sporulated oocyst, then sporozoites are released by the grinding activity of the gizzard and penetrate the cells of the intestinal mucosa. This begins the asexual cycle of development called schizogony. Next comes the sexual phase, resulting in the release of oocysts in the bird's feces. The entire process takes approximately 7 days.2 By day 14, after initial infection, the production of oocysts usually is diminishing and ceases around 18 to 20 days. Most damage to the intestine occurs early in the parasite's life cycle, during schizogony.
 |
Eimeria species
The Eimeria species that affect chickens
are E. acervulina, E. maxima, E.
tenella and E. necatrix as well as E.
brunetti, E. praecox and E. mivati. Those
of particular importance to breeder pullets
and roosters are E. acervulina, E.
maxima, E. tenella and E. necatrix.
The two species that cause intestinal
hemorrhage and pullet death are E.
tenella and E. necatrix. E. tenella is also
the species that causes bloody droppings,
along with mortality; it is often
the easiest for flock supervisors to recognize
due to the characteristic bloodfilled
ceca of dead pullets.
E. necatrix usually does not cause
many problems until after 6 weeks of
age because it does not compete as
well against other coccidia. This means
that mortality caused by E. necatrix usually
begins around 7 to 9 weeks of age;
necropsy signs of white/red or
white/black (salt and pepper) are seen
in the mid intestine. It is also important
to know that E. necatrix is the least
immunogenic of the chicken coccidia.
This will become more important when
we discuss control and immune
response.
Perhaps the more economically
important coccidia species the world
over for pullet and rooster frame size
and body uniformity are those that do
not cause death but have an impact on
the bird's ability to absorb nutrients
from feed. The most commonly recognized
are E. acervulina and E. maxima.
The signs of these two species are not
as obvious, so their effects are often
overlooked until it is too late.
Therefore, it is important to necropsy a
few birds during the first 3 to 4 weeks
of a pullet flock's life to determine levels
of these two types of coccidia. E.
acervulina will cause white stripes in
the duodenum, while E. maxima may
cause a ballooning of the intestine with
orange mucus in the lumen.2
There are three species of coccidia
that are difficult to identify in pullets
when performing a routine necropsy.
These are E. mitis, E. praecox, and E.
brunetti.
E. mitis is normally found in the
lower small intestine and produces
rather indistinct lesions. E. praecox also
does not have prominent lesions and is
also often missed at necropsy. Most of
the infection by E. praecox is in the duodenum
and may result in pinpoint hemorrhages.
E. brunetti can also affect the
lower small intestine, usually around
the yolk stalk. It does not produce any
recognizable gross lesions. All three of
these species are generally only diagnosed
by microscopy.2
Coccidiosis control
Control of coccidiosis in breeder pullets
and roosters can be summarized in just
one word: immunity. It does not matter
if we are using a drug or vaccine: in
both instances, the goal is to allow hens
to develop life-long, lasting immunity
to coccidia by 12 weeks of age.3 To
accomplish this goal, we must look for
ways to maximize the immune
response without causing a negative
impact on the birds' frame size and uniformity.
Factors that affect the development
of immunity are management conditions
such as litter moisture, partial vs.
full-house brooding and a feed restriction
regimen (skip-a-day feeding). We
also need to be aware of other disease
or live vaccine challenges, particularly
those that directly impact the immune
response, such as infectious bursal disease
virus, Marek's disease, chicken
anemia virus and exposure to mycotoxins
in feed.
Chemotherapy.
A variety of drugs will allow enough coccidia to complete
their life cycle for development of an
adequate immune response in the pullet/
rooster. We often refer to this partial
suppression as "leakage."
Anticoccidal drugs are broken into
two broad classes: chemicals and
ionophores. Chemical anticoccidial
drugs that can be used to allow immunity
to develop (leakage) in replacement
breeders are amprolium, amproli-
um with ethopabate, zoalene and clopidol
in the US. However, in Europe,
many of these products are no longer
available.
Amprolium has been used on many
pullet farms for about 40 years, so there
is a significant level of coccidia that
have become resistant to this drug.
Amprolium is very good against the
hemorrhage-producing coccidia E.
tenella and E. necatrix and it has some
activity against E. maxima. When you
add ethopabate, you broaden effectiveness
to include control of E. acervulina.4
 |
Clopidol and zoalene are very safe for
use in pullets, but resistance develops
quickly. Neither of these drugs has
been used extensively so they may be a
good choice for control of coccidiosis
in replacement breeders.
The ionophore anticoccidial drugs,
such as monensin and salinomycin, are
effective against all of the Eimeria
species of concern in replacement pullets/
roosters. Because they are coccidiocidal,
they are used at lower doses
than in broilers to allow immunity to
develop.
Vaccination.
Resistance to any anticoccidial
drug develops on a farm with
continuous use of the drug, which
selects for those Eimeria that can survive.
Over time, the proportion of coccidia
that are resistant to the drugs
increases and the result is the development
of more severe lesions, which can
lead to poor uniformity and adversely
affect frame size. The alternative is to
rotate from the anticoccidial drug to a
live vaccine containing drug-sensitive
strains.
Coccidiosis vaccines have two
advantages. Over time, the farm's population
of coccidia reverts back to being
sensitive to the anticoccidial drug while
the birds are vaccinated.5 The vaccine
also provides a "controlled exposure."
This means you know exactly when the
birds should experience the greatest
amount of coccidia lesions, which produce
immunity. In other words, you
know when to keep a close watch and
provide support to the birds if needed.
This can be especially important in pullet
flocks that are reared in concrete
floored houses or on new litter,
because exposure may be delayed for
these birds. The key to vaccination is
that it provides all the coccidia species
earlier in the bird's life than is seen with
natural exposure and results in a more
uniform development of immunity.
There are two types of live coccidia
vaccines available worldwide: attenuated
vaccines and controlled exposure
vaccines. In the United States, only controlled
exposure vaccines are available.
This means that the coccidia vaccine
given at one day of age are nonattenuated
sporulated oocysts and the birds,
when vaccinated at 1 day of age, will
experience peak oocyst production at
about 7 to 10 days of age until about 28
days.6 Outside the US, there are two
attenuated strains available for use in
pullets. Both products produce a good
immune response and may also produce
fewer lesions.
It should be remembered that pullets
will experience a reaction to E.
necatrix later (6-12 weeks of age) than
is seen with the other coccidia because
E. necatrix is less immunogenic. This
more predictable reaction to E. necatrix
is another advantage to using coccidial
vaccines because the reaction also normally
occurs very uniformly and earlier
than is seen with anticoccidial drug
programs.
Keep in mind that death and bloody
droppings are not the only signs to
watch for during this period of "vaccine
reaction." Reaction to E. acervulina and
E. maxima can affect the pullet's ability
to absorb vitamins, especially fat-soluble
A, D, E and K. This is one reason
that rickets may develop and that an
increase may be seen in leg problems
at around 4 weeks of age. One solution
is to routinely add vitamins to the pullets
drinking water during the vaccine
reaction period.
Treatment
Treatment may be necessary for various
reasons. One such scenario occurs
when the anticoccidial drug program
begins to fail due to an increase in the
number of resistant oocysts. Another
scenario occurs when there is an
extreme challenge before immunity has
had time to develop as might occur if
the litter becomes wet or there is a
delay in the development of immunity
after vaccination. Remember, the
choice to treat may slow or even stop
the development of the immune
response, so the flock must be watched
closely in the future for further coccidiosis.
It should be noted that routine
administration of anticoccidial medication
to vaccinated flocks can slow or
stop the development of immunity.
Therefore, good flock supervision, routine
necropsy of mortality and treatment
only when necessary is advisable.
In the US, our choice of drugs to
treat in the drinking water is limited to
amprolium and the sulfa drugs such as
sulfaquinoxoline or sulfadimethoxine.
In addition to these drugs, toltrazuril,
available in several countries outside
the United States, has been a very
effective medication. It is important to
identify the Eimeria species most affecting
pullets/roosters, because amprolium
is more effective against E. tenella
and E. necatrix (hemorrhage producers)
and sulfa drugs work best against
E. acervulina and E. maxima.4
Summary
Today's replacement breeders are far
more feed-efficient and grow more rapidly
than they did 25 years ago.
Consequently, we must do a better job
managing the development of immunity
to coccidia if we are to minimize the
impact on skeletal frame size and body
weight uniformity. There are several
options available to help birds develop
lifelong immunity, ranging from anticoccidial
drugs to vaccines.
Whichever method is used, birds
must still be closely monitored for signs
of excessive coccidial damage. If we
don't do a good job managing our
"cocci program," we may significantly
affect both egg production in hens and
fertility in roosters.
References
1 Boren B. Does frame size matter? In:
Proceedings of the Arkansas Poultry Symposium,
Springdale, AR. April 9-10:15-22,
2002.
2 McDougald LR and Reid WM. Coccidiosis.
In: Diseases of Poultry, 10th edition,
B.W. Calnek, ed. Iowa State Press, Ames,
Iowa. pp. 865-883, 1997.
3 Bafundo KW. Managing coccidiosis in
broiler breeder replacements. World Poultry,
Vol. 7, No. 9:30, 1991.
4 Gill G. Anticoccidials versus Eimeria:
Managing drug resistance. Feed Management,
Vol. 49, No. 7:12-17, 1998.
5 Chapman HD, Cherry TE, and Quiroz MA.
Sensitivity of field isolates of Eimeria to
anticoccidial drugs following the use of
Coccivac in broilers. In: Proceedings of the
Forty-fifth Western Poultry Disease
Conference, Cancun, Mexico. May 1-5:107-
108, 1996.
6 Stiff MI and Bafundo KW. Development of
immunity in broilers continuously exposed
to Eimeria sp. Avian Diseases. 37:295-301,
1993.
Source: CocciForum Issue No.6, Schering-Plough Animal Health.







